It is rare case and real tough one. A clue : concentrate inside the pericardial space. (36, year old man , asymptomatic)
Know the diagnosis
Source and reference & Courtesy
Comment : Most curious , who stopped it from entering LV ?
Posted in Uncategorized, tagged bullet injuries of heart, echocardiography, gun shot injury of heart, miracles in clinical cardiology, pericardial disease, rare cases in cardiology, rare foreign body in heart on January 27, 2026|
It is rare case and real tough one. A clue : concentrate inside the pericardial space. (36, year old man , asymptomatic)
Know the diagnosis
Source and reference & Courtesy
Comment : Most curious , who stopped it from entering LV ?
Posted in Uncategorized, tagged doac for lv mural thrombus, doac vs oac, efficacy of doac in lv thrombus, rivaroxaban, warfarin vs doacs on January 21, 2026|
Does DOACs trying to push over warfarin in post STEMI LV mural thrombus ?
Yes , it seems so. Old must give way for new, even if it is gold. We need to go with the trend. Only thing, we need to prove is, whether the new ones are safe . Need not bother much about efficacy. Some one, will somehow, prove the new ones , non-Inferior to the best available drug as on date.
Every experienced cardiologist would know, Warfarin is many-fold more effective. But, still NOACs are coming in a big way. With the help of big trials, NOACs will ultimately replace the much more dangerous, bleed-prone Warfarin. One more reason we might embrace the inferior drug is the clinical inertia for vigilant INR monitoring.
What does the evidence say ?
Read : Ref 2
The future of medicine will be fight between quality & ( Intention ) of evidence and experience
Caution : Personal opinion
It would be science at its best, when a less potent drug, that requires zero monitoring, will replace a more potent and established OAC. It doesn’t require brilliant brains, to realize a drug which has less bleeding risk, is going to be less effective in getting rid of thrombus as well. (The trial in Ref 2 proved otherwise)
Only issue with warfarin is, we need to be a little more careful. Fortunately, its usage in LV mural thrombi is not likely to be permanent. (as in prosthetic valve)
Reference
AHA comprehensive review on LV thrombus management
A new pre specified -meta-analysis (I don’t know what it means) from a popular Journal EuroIntervention shows a strong evidence in favor for DOACS
In the above study, Warfarin trails behind Rivaroxaban by a huge 20% margin in it’s ability to successfully remove LV thrombus.

TThere is a surprise in the conclusion : Why do the authors, hesitate to commit DOACS are superior? Something to think about.

Posted in Uncategorized, tagged best medical quotes, bmj, hippocrates, jama dr s venkatesan sangareddi, lancet, medical ethics, nejm, osler willam, pateint care vs science, practical bio ethcis, primum non nocere, science and ethics on January 14, 2026|
Posted in Uncategorized, tagged BASILICA, CATHEDRAL, innovations in cardiology, NEO CORONARY OSTIUM, PERCUTANEOUS cabg, TACKLING CORONARY OBSTRUCTION AFTER TAVI, TAVR, UNICORN, VECTOR on January 14, 2026|
Innovation and Interventional cardiology are like Inseparable conjoined kids. When a mind blowing idea shakes hands with unprecedented technology, supported by an accommodating industry, such breakthroughs happen.
First In human : Creation of neo-coronary ostium & by pass stent to LAD
Here is an ultimate intervention, called the VECTOR procedure, that creates a tunneling pathway outside the regular anatomical pathways, like laying a road or metro tunnel under a mountain or river. (Ref 1 Circulation Cardiovascular Intervention 2026)
Combining the expertise of the retrograde approach of CTO and covered stent technology, a neo coronary ostium and an artificial artery is created that connects the ascending aorta and LAD, burrowing through the epicardium, myocardium, and pericardial plane.
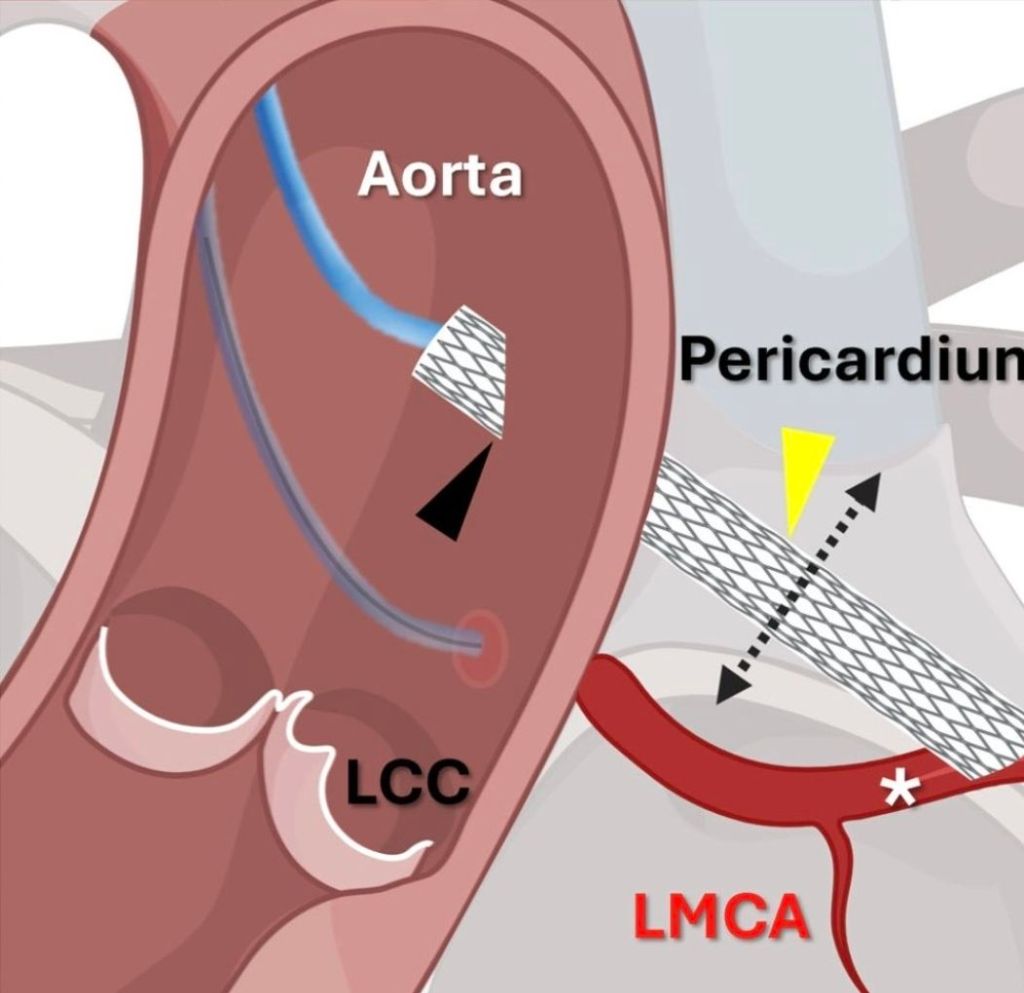
VECTOR procedure : Ventriculo-coronary transcatheter outward navigation and reentry (VECTOR)
1.Who is the patient ?
A very high risk post TAVR, who had a blocked his left coronary ostium. VECTOR was attempted in lieu of CABG, which probably he was not eligible.
2.Where it was done ?
I think it was done at Emory , the same place Andreas Gruentzig, show cased his magnum opus.
(It is a combined innovation from four institutes Structural Heart and Valve Center, Emory University Hospital, Atlanta, National Heart, Lung and Blood Institute, National Institutes of Health, Bethesda, St Francis Hospital and Heart Center, NY, MedStar Washington Hospital Center and Georgetown University, Washington, DC0
3.What are possible percutaneous solutions to prevent coronary ostial obstruction ?
1.BASILICA (Bioprosthetic or native Aortic Scallop Intentional Laceration to prevent Iatrogenic Coronary Artery obstruction)
2.UNICORN (Undermining Iatrogenic Coronary Obstruction With Radiofrequency Needle)
3.CATHEDRAL (CATHeter Electrosurgical Debulking and RemovAL)
The VECTOR procedure , first in human human was attempted
4.Now, what are challenges ? Will such procedure stand the test of time ?
The key question is, (apart from complexities and complications of the VECTOR procedure) how the covered stent will seal the peri-anastomotic site both in the aorta and coronary end?
5.Is this procedure possible in a non-TAVR situation in complex ostial and left main disease?
Time will tell
6.A venous and hepatic analogy
I don’t know this comparison is correct. VECTOR procedure has some similarities with the TIPS procedure done as percutaneous portal vein to IVC connection through the hepatic tissue planes (Vignali C, TIPS with expanded polytetrafluoroethylene-covered stent: results of an Italian multicenter study. AJR Am J Roentgenol. 2005 )
Final message
If we can, somehow make the procedure simple with smaller hardware, circumventing VECTOR will be an ultimate victory for mankind and sure to stand tall in the pinnacles of glory.
However , when adopting new technologies “Always be ready, to sacrifice science , if you think it would interfere with patient well being”.
Reference
Posted in Uncategorized, tagged cardio renal syndrome, heaptic and reanl congestion, ivc hypertension in cardio renal syndrome, renal vensous hypertension, Role of right ventricle in cardio renal syndrome, rv dysfunction in cardio renal syndrome on January 11, 2026|
Cardio-renal syndrome (CRS) , in reality seems to be , a vague clinical entity, defined and classified into 5 types essentially for our convenience and simplicity. This classification do not contribute to any pathophysiological insights . The fact of the matter is, every patient with heart failure (both acute and chronic) has some amount of renal compromise & vice versa. The link between LV failure and renal compromise is more discussed and documented in the literature than the influence of the right ventricle.
Now evidence is emerging, like hepatic congestion , renal venous congestion also play an important role in compromising renal function. Traditionally, we aim to increase the GFR, by improving renal blood flow, but the real problem may lie in clearing the exit paths of the renal venous system.
A mini review on the topic
Right ventricular (RV) dysfunction in chronic heart failure (HF) contributes to renal dysfunction beyond reduced forward cardiac output, primarily through backward venous congestion elevating inferior vena cava (IVC) pressure and renal venous hypertension. This congestion transmits high central venous pressure (CVP) to renal veins, increasing interstitial pressure, compressing tubules, and impairing glomerular filtration rate (GFR) independent of arterial underperfusion. Studies confirm IVC dilatation and CVP >8 mmHg correlate with worsened renal function, reversible by decongestion.
Elevated RV filling pressures cause systemic congestion, reducing trans-renal perfusion pressure (mean arterial pressure minus CVP) and promoting renal parenchymal hypoxia. Animal models show renal venous pressure elevation decreases urine output and GFR more than equivalent arterial hypotension, via tubulo-glomerular feedback disruption and renin-angiotensin-aldosterone system activation. Human data from acute decompensated HF link high RAP/CVP to serum creatinine rise, with decongestion improving GFR in RV failure.
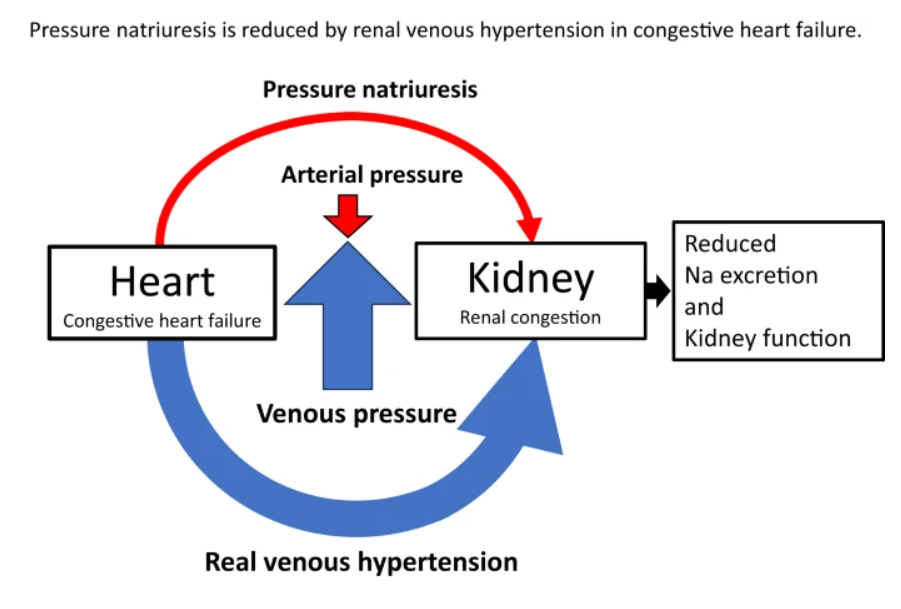
RV-driven congestion activates intrarenal mechanoreceptors, triggering sympathetic vasoconstriction and hepatorenal/splenorenal reflexes that exacerbate sodium retention. Splanchnic pooling limits venous capacitance, amplifying intra-abdominal pressure and gut-derived inflammation (e.g., LPS translocation), fostering a proinflammatory milieu worsening cardiorenal interactions. Ventricular interdependence from RV dilation further impairs left ventricular output, compounding renal hypoperfusion.
The AHA Scientific Statement on Cardiorenal Syndrome emphasizes venous hypertension over hypoperfusion in CRS type1 and 2 stressing the importance of ESCAPE/DOSE trial results on diuretic-guided decongestion.
Clinical Implication
Major implication is the role of optimizing diuretic dose in HF, which can reduce renal venous congestion . Treatment strategies aiming to improve pulmonary hypertension and RV function seems important as well.
Final message
If we need to manage cardio-renal syndrome effectively, we need to pull both Cardiologists and Nephrologists to sit in a combined consultation (a difficult proposition in current times), and review patient data, optimizing the strengths and weaknesses of all pillars of heart failure drugs, and (more importantly) fine-tuning the important fifth pillar, the diuretics*.
*For some reason, the literature and cardiology community refuses to admit a blatant truth : Diuretics are life-saving in heart failure.
Further reading
How to treat Congestive Cardio-Renal failure ?

Posted in Uncategorized, tagged coapt study, eccentric cs concentric mr, jacc, lancet, lub dub, macr, Mechanism of mitral regurgitation, mitra clip, mitral annulus, nejm, role of stethoscope in current era, scai, TAVR, teer, tethering cs closing forces in mitral valve on January 3, 2026|
Preamble : The Lubs & Dubs
The lubs and dubs, along with some added sounds are the only language, the heart can speak in health and distress. It’s a worrying story altogether, gradually many of us are becoming “cardiac illiterates” as we struggle to read , its gentle communication. it is not our fault. Stethoscopes are reduced to become a social marker of being a doctor. We may excuse ourselves, even if we can’t differentiate a systolic from diastolic murmur, after all, hand held echo machines, instantly tell the diagnosis.
( After reading this article, fellows are expected to understand why the first heart sound in MR (ie the lubs,) are mostly soft, some times normal or even loud in certain conditions)
Now, let us go to the mitral valve dynamics
How many of us are aware, there is a big science of physics and biology operating when the mitral valve perfectly closes at the level of the annulus, with each systole , balancing different sets of known and unknown forces.
In this article, we will see how these two sets of forces mitral valve tethering and closing forces balance out each other to seal the mitral valve and what happens when the forces begin to fight each other.
Balance of Tethering and Closing Forces in Mitral Valve Coaptation
The mitral valve (MV) coaptation refers to the edge-to-edge apposition of the anterior and posterior leaflets during systole, ensuring a competent seal to prevent regurgitation. This process is governed by a delicate balance between tethering forces (which restrain leaflet motion to prevent prolapse into the left atrium) and closing forces (which approximate the leaflets for sealing).
MV mechanics exhibit several counterintuitive paradoxes, where adaptive or dysfunctional responses lead to outcomes opposite to expectations. These highlight the interplay of geometry, contractility, and force transmission:
This article clearly tells us that the forces acting on the mitral valve apparatus are so complex. The conceptual model of tethering and closing forces may be oversimplified. There are variable interactions between them. More importantly, the atrial forces also influence and intrude into these forces. Realize that MV competence is not just about force magnitude but their vectorial distribution and timing, often amplified by LV geometry changes.
Final message
As cardiologists and surgeons, we must realize the fact, how important it is to analyze both anatomy and the physiological impact when we rush to clip, cut, or repair it with annuloplasty and subvalvular interventions.
*Sometimes, it might even be tempting to do mitral valve replacement, even when it is not indicated, because we need not bother about all these dizzy mechanics and physics of MR jet forces.
Posted in Uncategorized on January 1, 2026|
As we enter, another customary happy “New year” , a lingering “Old wish” remains largely unfulfilled. Let us try to return, to our forgotten home space, called patient’s bed side . Shall get Immersed in history taking , Intuitive clinical examination, and master the art of listening to our patient’s heart with our own ears. Investigations can wait unless it is a dire emergency.
Too often today, we bypass these foundations, relying blindly on Images, echocardiograms, angiograms, a deluge of scans, , multi-modality algorithms ,AI predictions. We have also become greedy servants to technology commerce , and increasingly intoxicating science as well. Let us not insist on investigations , driven by peer pressures or pride, in the process losing common sense in a flood of data.
Let us reclaim the intellect, that taught us listening and understanding to the patients symptoms (with kindness) is the highest form of Investigation .
Coming to scientific research, grow courage to question, debate , that ultimately would simplify complex problems .
Finally, seek the truth, which often hides behind the distorted evidence base and obsessive compulsive protocols.
Welcome to new year 2026
Greeting & regards from Chennai.
Dr. S.Venkatesan
Posted in Uncategorized, tagged acc, aortic filter, circualtion research, embol x, esc, innovations in cardiology, ivc filter, laaa occluder, lancet jacc interventions, nejm, PAA-PMF, permanent aortic filter, scai, sentinel device, stroke prevention, TAVR, triguard on December 24, 2025|
“Every Interventional Cardiologist, realistically, need to be a preventive neurologist too!”
The concept a permanent ascending aortic porous membrane filter (PAA-PMF) is an extrapolation of the idea of mechanical thrombus capture, as proven by IVC filters for venous embolism prevention . Also we do have and temporary intra-aortic filters like Sentinel , Embol-X for arterial particulate capture.
The PAA-PMF would feature a self-expanding nitinol frame, with a fully porous head end. The device can be heparin-coated polyester or polyurethane mesh membrane, deployable via 12-14 Fr femoral sheath, similar to IVC filter designs but should be optimized for aortic pressures. Suggested pore size of 100-125 μm targets >100 μm emboli, akin to Embol-X filtration efficacy in capturing 95% of particulates (atheroma, fibrin) during aortic declamping. The essential requirement is that the porous membrane should not create an impedance gradient. How feasible it is, to be tested. Conical shape, the radial force will ensure good ascending aortic wall apposition.
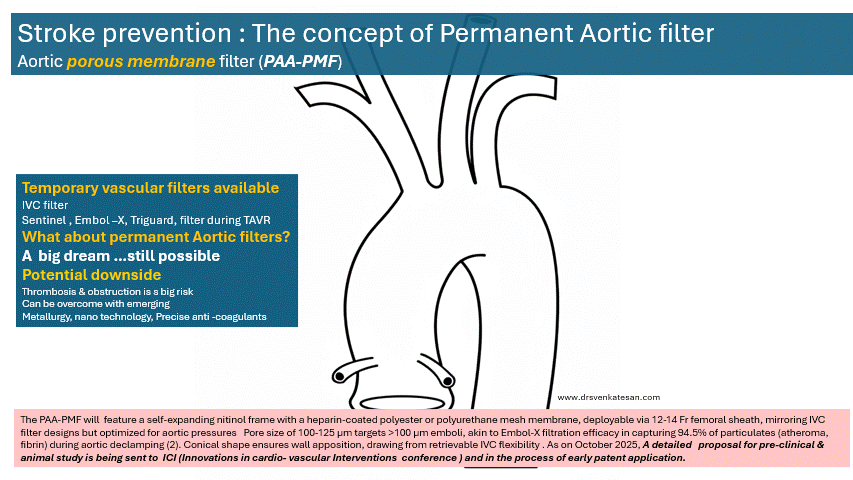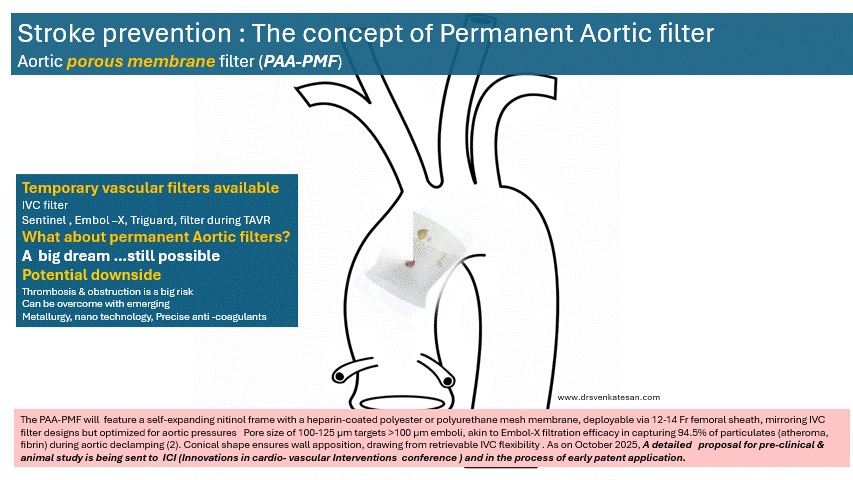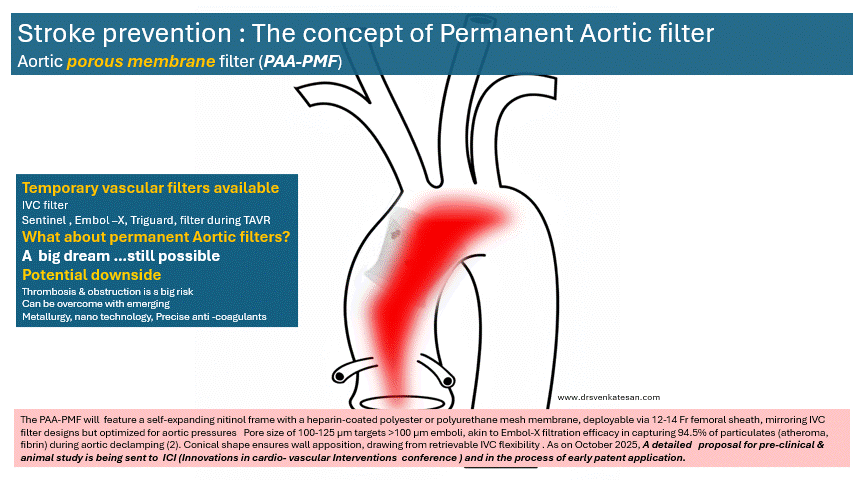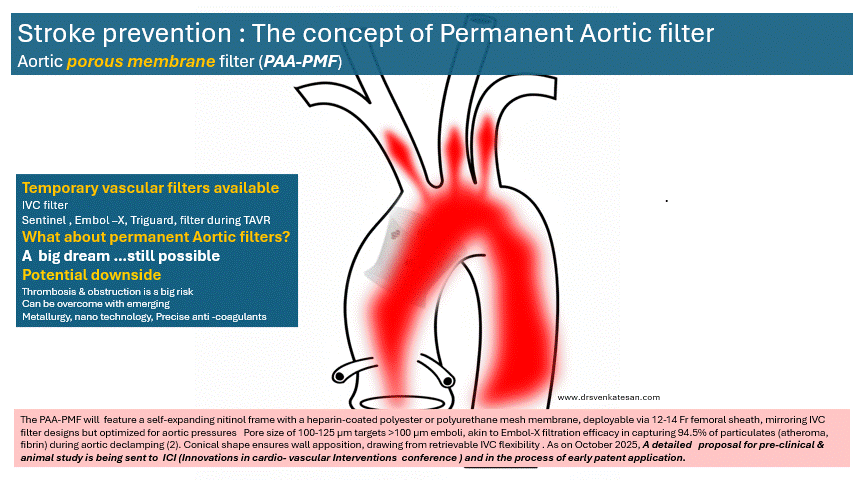
Site of placement is critical. Proximal ascending aorta, 2-3 cm distal to sinotubular junction/proximal to brachiocephalic trunk, as in Embol-X for maximal cardiac/aortic debris interception without coronary/arch compromis
(Only in patients with very high risk of cardioembolic stroke)
1.Chronic stroke reduction in patients with MVR/AVR/TAVR/MAVR
2.High-risk mobile LV mural thrombus
3.Chronic AF with visible and invisible clots in LA
4..High-risk procoagulant conditions with recurrent embolism
*Occlusion and hemodynamic compromise is the most crucial issue. However, when compared to the incidence IVC filter clogging, the high pressure aortic flow is likely to self-wash the device (as happens in a prosthetic aortic valve)
Trapped emboli may enter into coronary circulation is a possibility. Putting a filter at ascending aorta precludes left heart catheterization.
*Migration , Hemolysis are other expected complications.
Intense anticoagulation would be required to prevent occlusion of the filte . (Still, stopping it temporarily doe not not increase the risk of stroke)
Final message : Is it Worth for a Preclinical trial ?
We do have temporary aortic filters. The concept of permanent or semi-permanent filters is largely theoretical, with potential risks being more than benefits. The device can take care of only cardio-aortic embolic stroke.
However, considering so many complex, risky intracardiac and intravascular devices being tested on a daily basis, it is not a big deal for the current generation of interventional cardiologists to try this.
More than our interventional appetite, we really need a device that prevents stroke in a permanent fashion. It is definitely worthy to do initial studies in a porcine model. Would be glad , if Edwards, Abbot or Medtronic and other new Innovators respond to this.
Posted in Uncategorized, tagged humanity in medicine, medcial ethics, science and medicine on December 24, 2025|
Posted in Uncategorized on December 21, 2025|
It would be silly to remind, it’s the same five liters of blood, that circulates across, both the arterial and venous system. But, its journey one away from the heart, and the other towards the heart are strikingly different. They are subjected to various hemodynamic forces, travels different terrains, at different speeds, thousands of kilometers of microvasculature along the cardiovascular highway, yet merging with each other every 15 seconds or so, at the pulmonary junction box. Have a look at the following images, to understand the distribution of the blood volume.
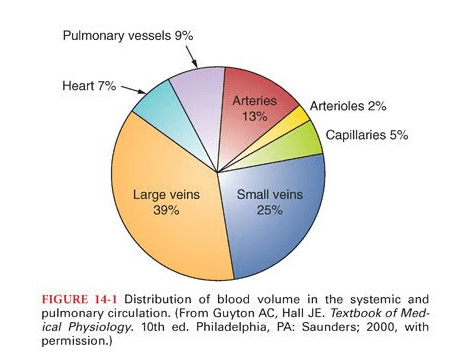

The first image is taken from the maverick physiologist Dr. Guyton’s textbook of physiology, and the second one from the equally famous Dr. Ganong’s. Both images depict the distribution of blood volume, the corresponding pressures, and velocity. Every cardiology fellow should recall these two images even in their sleep. Also mind, they circulate around the body, lifelong without clotting or bleeding, assisted by the right balance of pro and antithrombotic forces.
Why some of patient’s blood is more likely to get frozen ?
Logic would suggest venous thrombosis should be more prevalent than arterial thrombosis at any point of time and location. This is due to the slowness of the circulation and the enormous volume within the venous reservoir. But is this the clinical reality? It is indeed true, that incidence of minor venous thrombosis exceeds arterial thrombosis. Since venous thrombosis often gets lysed or get stuck in the lungs, it’s frequently under-recognized. Arterial thrombosis causes more damage in an important sense, as it leads to target organ ischemia.
Apart from hemodynamic factors, the 200 year old Virchow’s triad is very much alive. The vessel wall integrity, intrinsic defects in the coagulation and anticoagulant/fibrinolytic molecules, the genetic susceptibility are the important determining factors. The RBC and platelet behaviour too changes, in high and low pressure environments.
How to diagnose a patient who is in a procoagulant state?
The topic is so complex .Many things are still poorly understood. We should have a checklist of all systemic conditions that can cause increased risk of thrombus. We know pregnancy is inherently a procoagulant state, as is manifest or concealed malignancy.
What we normally do ?
It is very easy to tick the coagulation profile/panel slip and pass it on to the nursing staff. Some of us take another easy route, referring such patients to a rheumatologist for the risk-stratifying job. This is probably because we strongly believe SLE and connective tissue disorders are the first culprits.
I think we need to engage the hematologist more often because thrombosis is not only due to excess coagulation. It is also due to a lack of enough circulating anticoagulants. (As a cardiologist, sometimes I feel awkward. to call myself an expert of the circulatory system, with almost zero knowledge of how the blood clots or dissolves.) This article tries to differentiate the risk factors operative on the venous and arterial sides. It is only a gross attempt; many risk factors are invisible and are common between arterial and venous thrombus.
For a detailed analysis of Sydney criteria /ACR-EULAR (Reference 7 )
How to treat pro-coagulant state?
Fortunately, identifying the thrombosis prone patients is complex , but the treatment is fairly simple. We have only few options: Aspirin, Warfarin, and NOACs *We need to choose one of them. The general rule is aspirin doesn’t work much on the venous side. I don’t know how far this is really true. (It has something to do with the shearing force of platelets? ) However, in obstetrics, the placental circulation is full of low pressure venous plexus where Aspirin is used as a norm.
Between Warfarin and NOACs, there is absolutely no doubt Warfarin is the clear winner on the arterial side. Because of monitoring issues and fear of bleeding, we are compelled to switch to NOACs in many situations. Beware, think twice before prescribing NOAC for prophylaxis against arterial thrombus. The venous side does not have much difference in choice. *Heparin (& its glamor sibling LMWH) is a unique molecule, which has ability to work on both arterial and venous sides.
Postamble
The article doesn’t discuss the intra vascular metals, wires, devices, valves, pacemakers , related thrombosis. Here there is a known trigger. It is possible, they also influenced by the baseline factors of pro-coagulation discussed above.