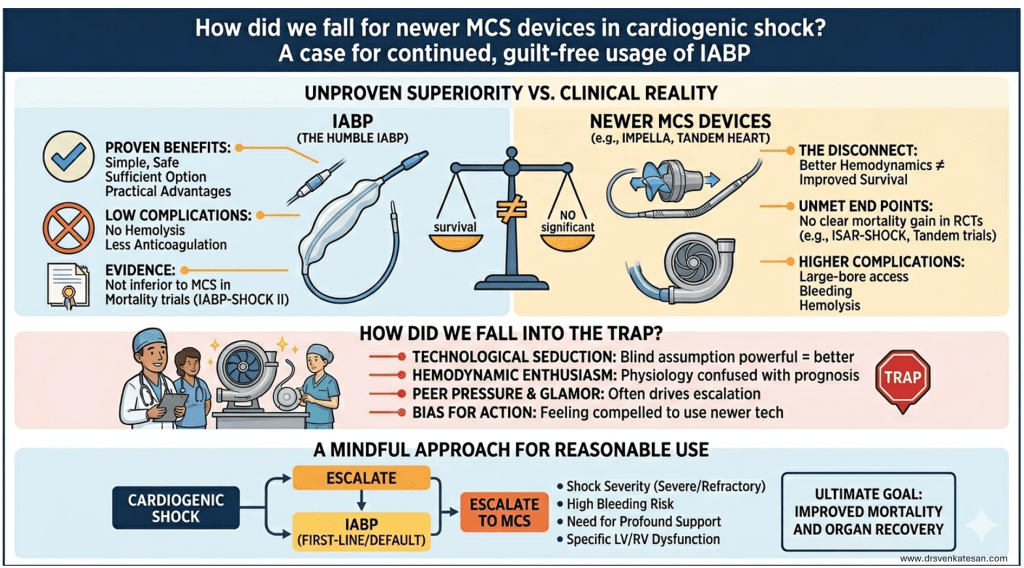
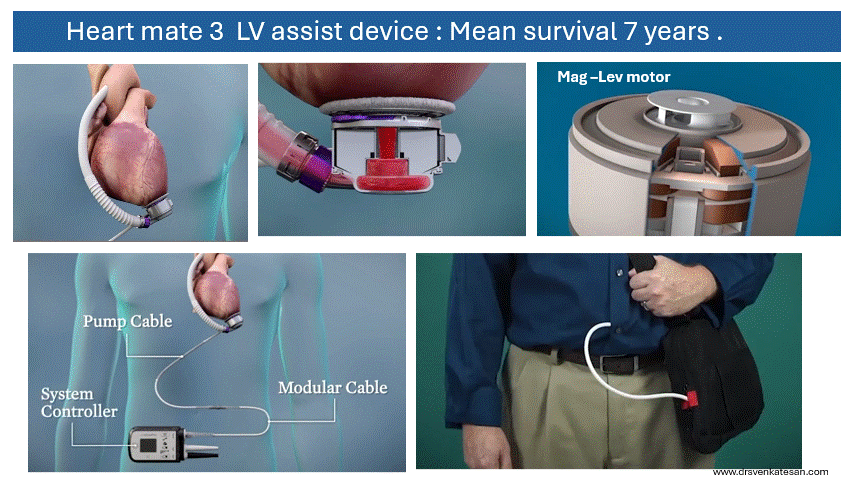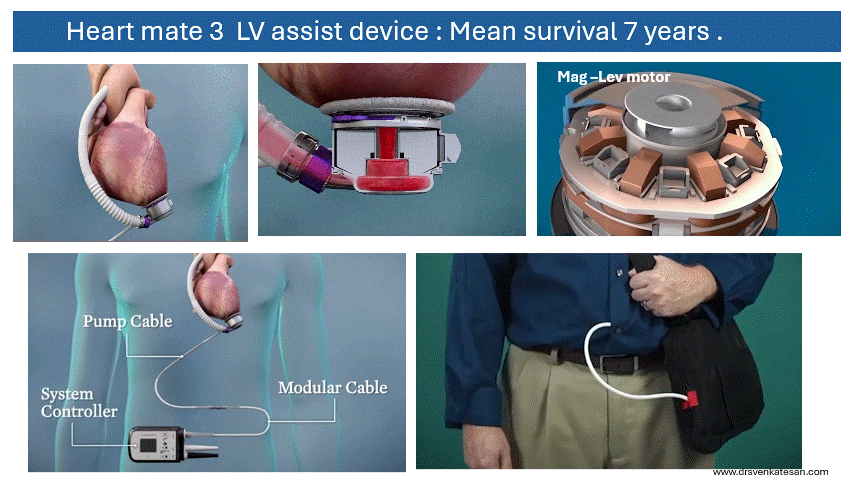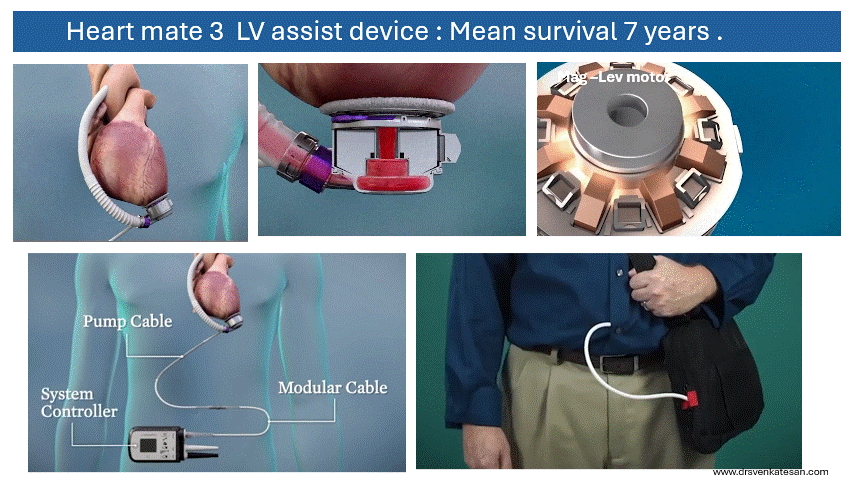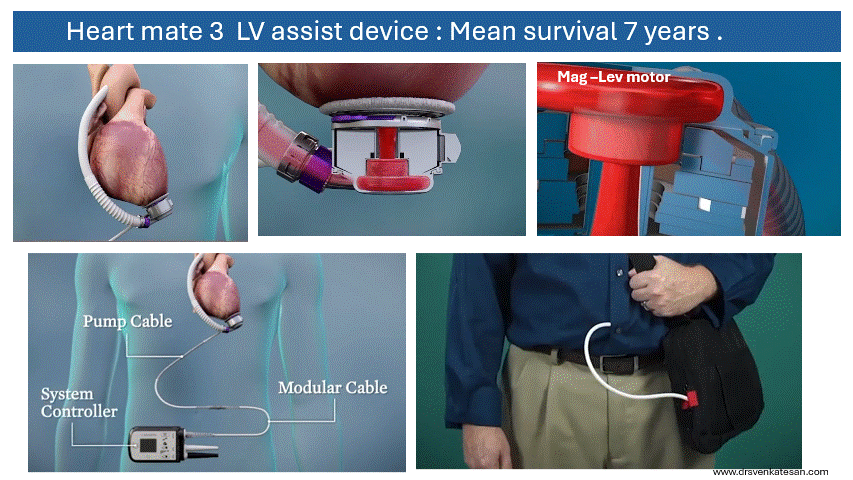
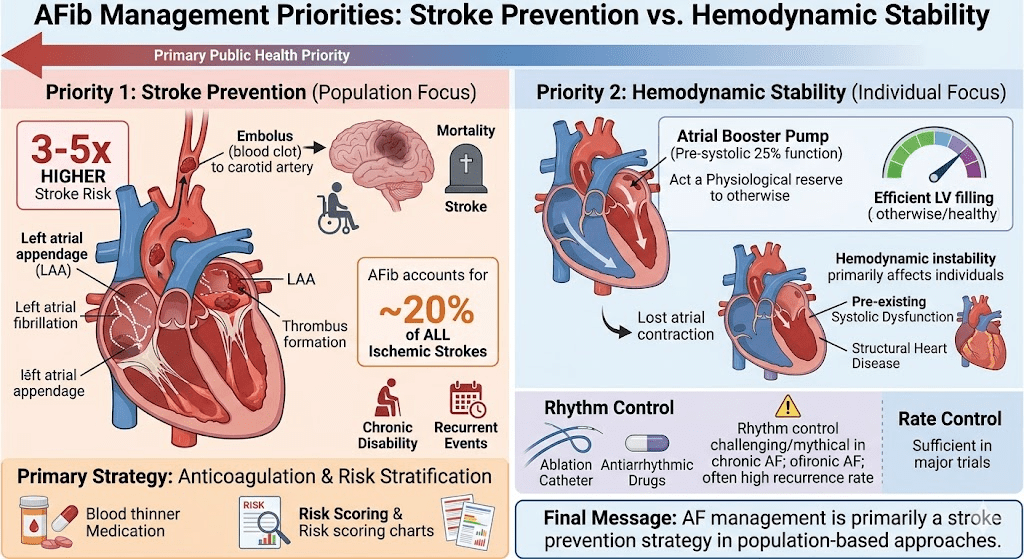
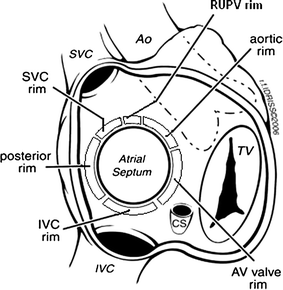
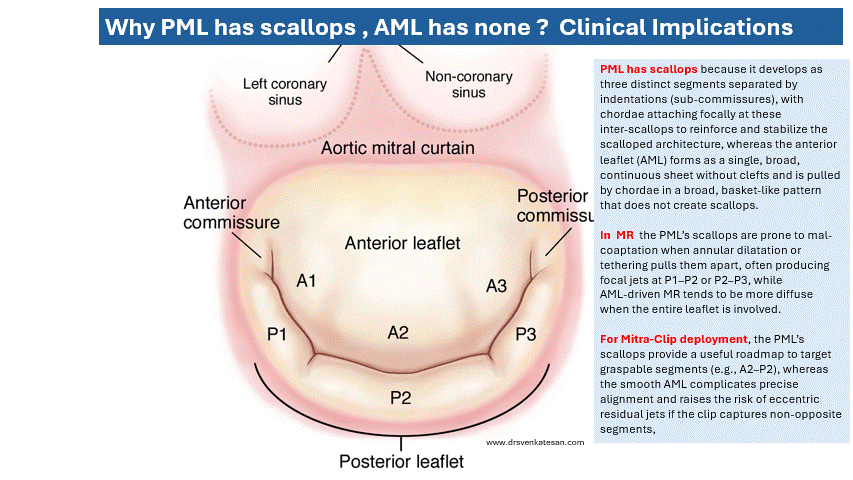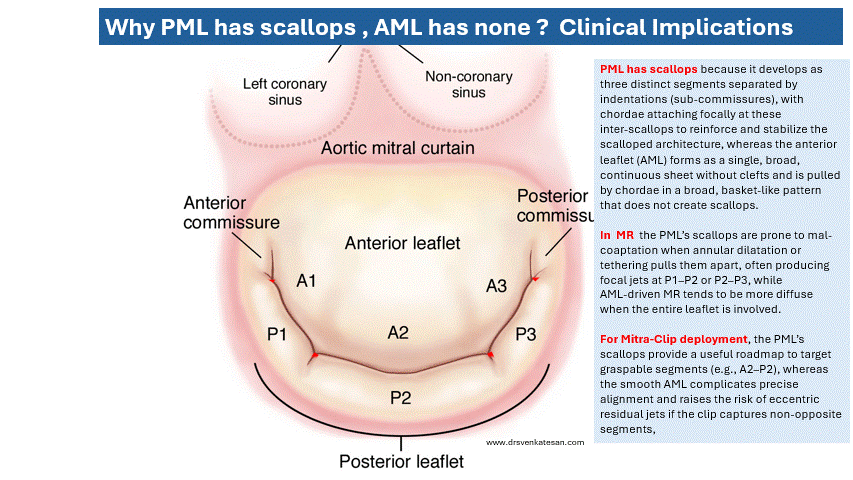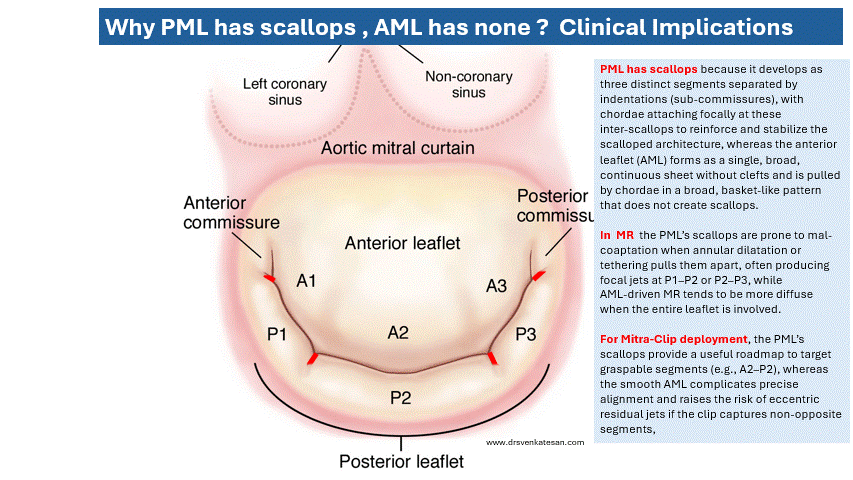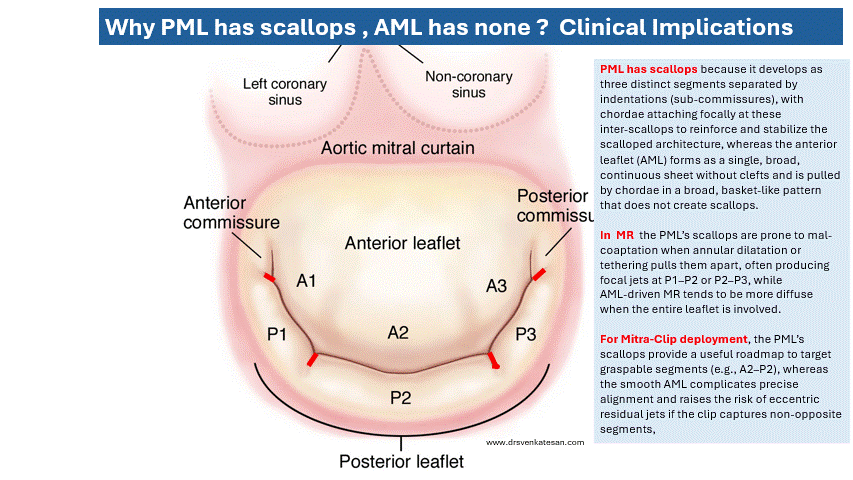
Some truths about True & False lumen :
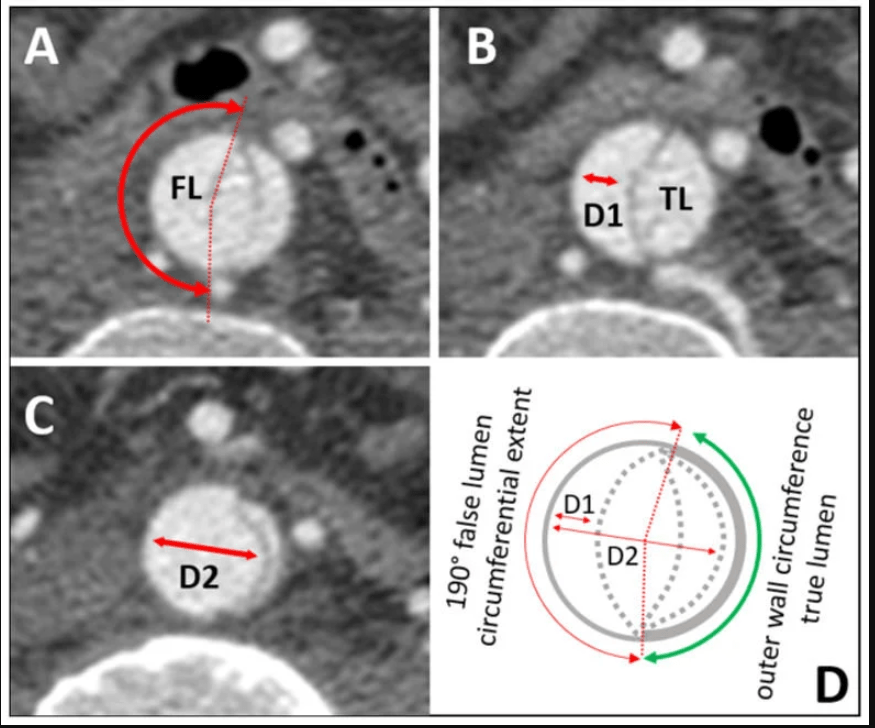
Aortic dissection splits the aorta into a true lumen (TL) and a false lumen (FL) at different planes of aortic media. True lumen often retains* all the three wall layers (intima, media, adventitia), providing structural stability. Conversely, the FL outer wall consists only of outer media and adventitia. This thin wall can be fragile. Persistent, un-thrombosed FL flow may maintain organ perfusion but it carries a risk of aneurysm or late rupture. The false lumen has less elastin, in long term more likely to to replaced with fibrosis. In spite of this , there are occasions were false lumen can by exploited for a good circulation. (Click here for more eloborate True vs false lumen difference)
Image source : Anna M H Sailer Circulation Cardiovascular Imaging 10(4):e005709
*A significant portion of wall of true lumen faces the false lumen .So technically true lumen also lacks three layers in its entire 360 degrees. But the risk of rupture is internal not in the aortic wall.A comforting truth about false lumen.The fragility of false lumen is highly variable, depending on the thickness of media .However, adventitia , the toughest layer of of Aorta is always there to limit the risk of rupture in the outer curvature.
Situations where false lumen Stenting is only way out.
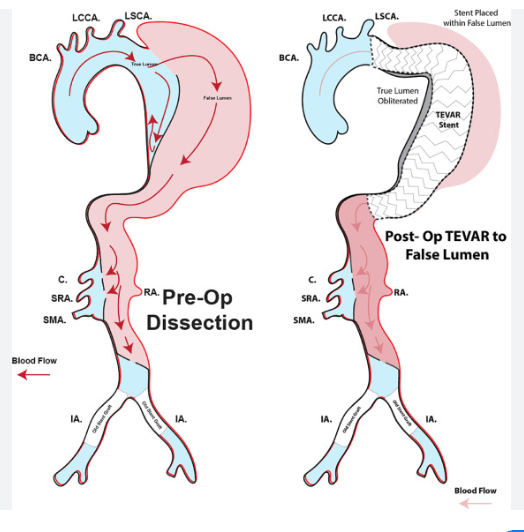
While standard endovascular repair aims to stent the TL and obliterate the FL, stenting the FL itself is utilized in two distinct scenarios:
- Intentional Organ Salvage: In chronic or severe dissections, vital branch arteries may arise solely from a dominant FL. If the TL is too crushed or severely calcified (or porcelain like ), we need to intentionally stent the FL to preserve critical organ perfusion.
- The STABILISE Technique: This aggressive strategy uses a TL stent followed by heavy-duty balloon inflation. The balloon deliberately breaks the internal dissection flap, obliterating the FL space and fusing both channels into a single, stable lumen.(Ref 2)
Inadvertent false lumen stenting
This is more common than we realise.Some times it is not never recognised ,if the entry and exit paths are clean. But more often it creates immediate hemodynamic issues.A guidewire can accidentally enter the dynamic FL . Deploying a stent here can catastrophically crush the TL, requiring immediate rescue fenestration or secondary TL stenting to restore flow.
Conclusion
Identifying entire morphology of true and false lumen is critical. Restoring true lumen flow, and trying to obliterate false lumen is the default strategy in majority of aortic dissections. However, it is good to recognise , false lumen stenting is not forbidden, if situations demand. It can be life-saving in some complex anatomy or acute organ malperfusion.
Reference
- Simring D, Raja J, Morgan-Rowe L, Hague J, Harris PL, Ivancev K. Placement of a branched stent graft into the false lumen of a chronic type B aortic dissection. J Vasc Surg. 2011 Dec;54(6):1784-7. doi: 10.1016/j.jvs.2011.05.053. Epub 2011 Jul 23. PMID: 21784607.
- Hofferberth SC, Nixon IK, Boston RC, McLachlan CS, Mossop PJ. Stent-assisted balloon-induced intimal disruption and relamination in aortic dissection repair: the STABILISE concept. J Thorac Cardiovasc Surg. 2014;147(4):1240-1245. Available from: The Journal of Thoracic and Cardiovascular Surgery or via PubMed Central.
- Resch TA, Kölbel T, Dias NV, Rohlffs F, Carpenter SW, Heidemann F, et al. Intentional false lumen stenting for complex chronic aortic dissection: te