Embryology
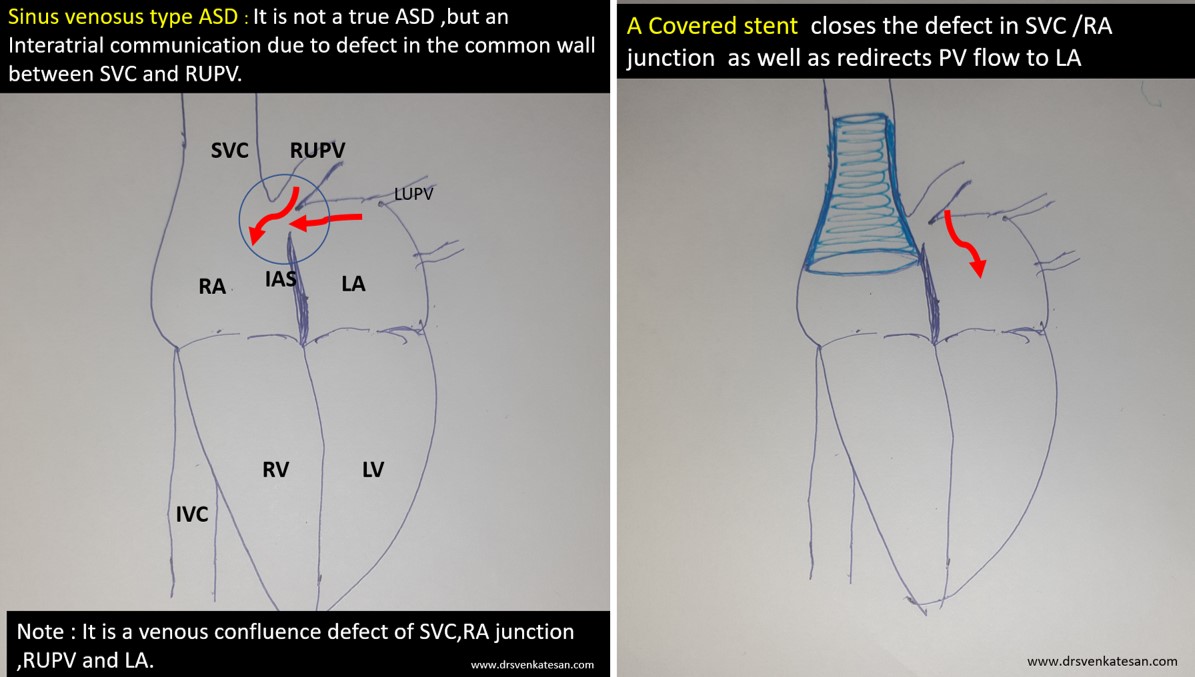
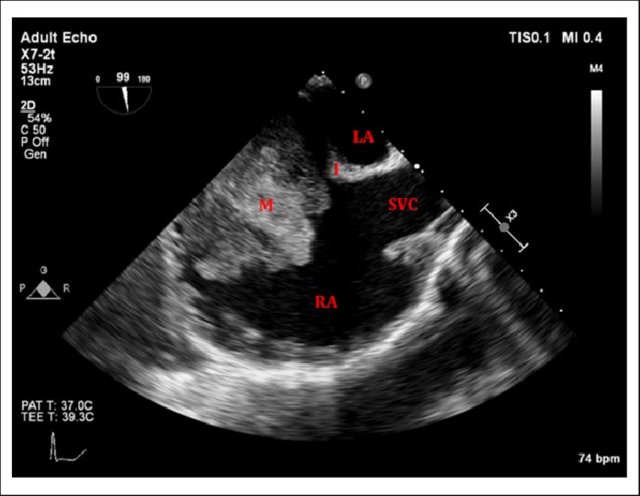
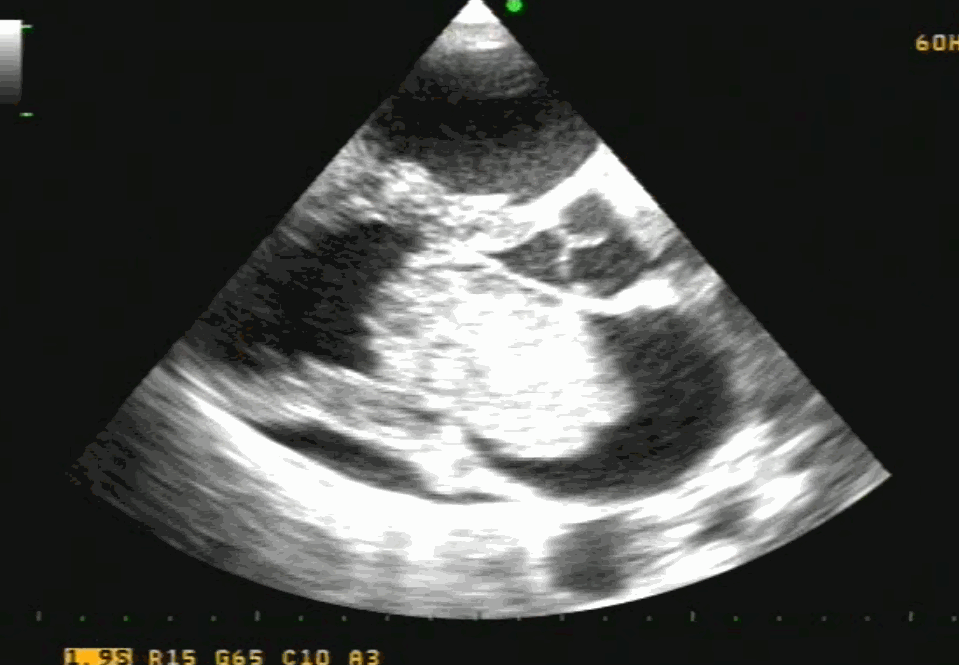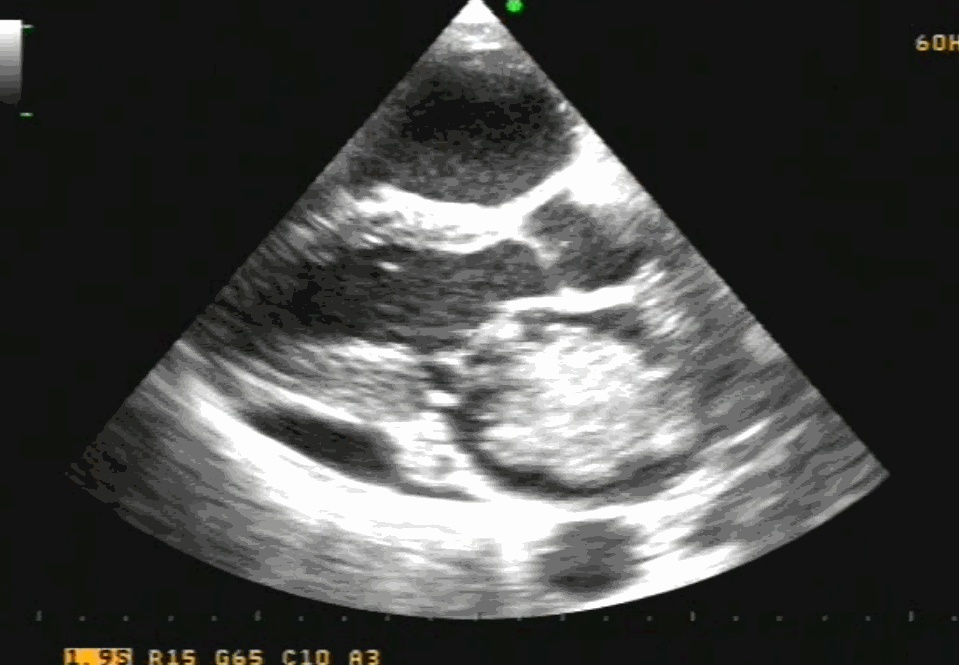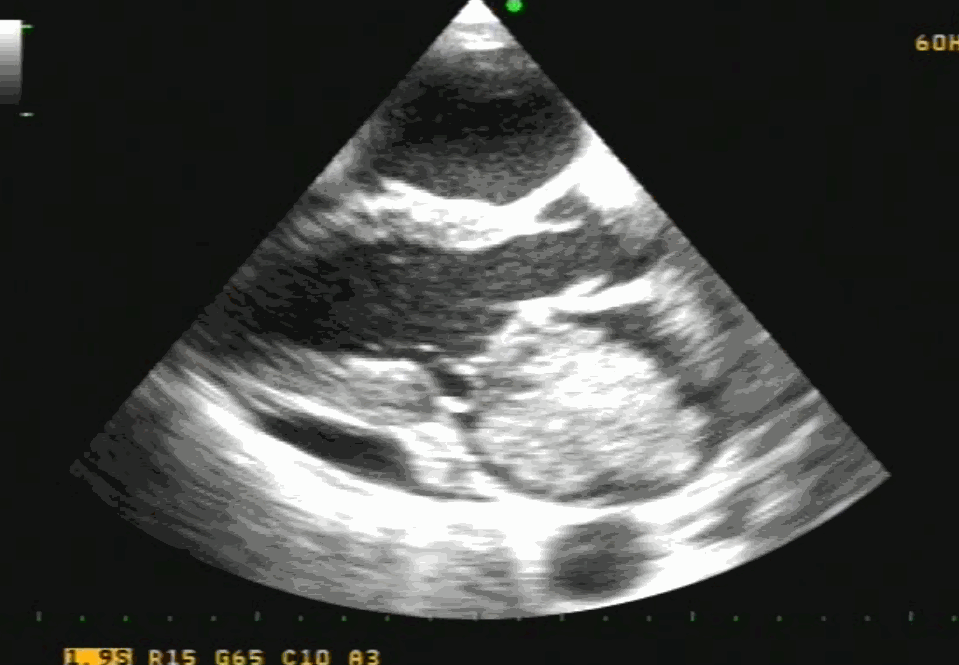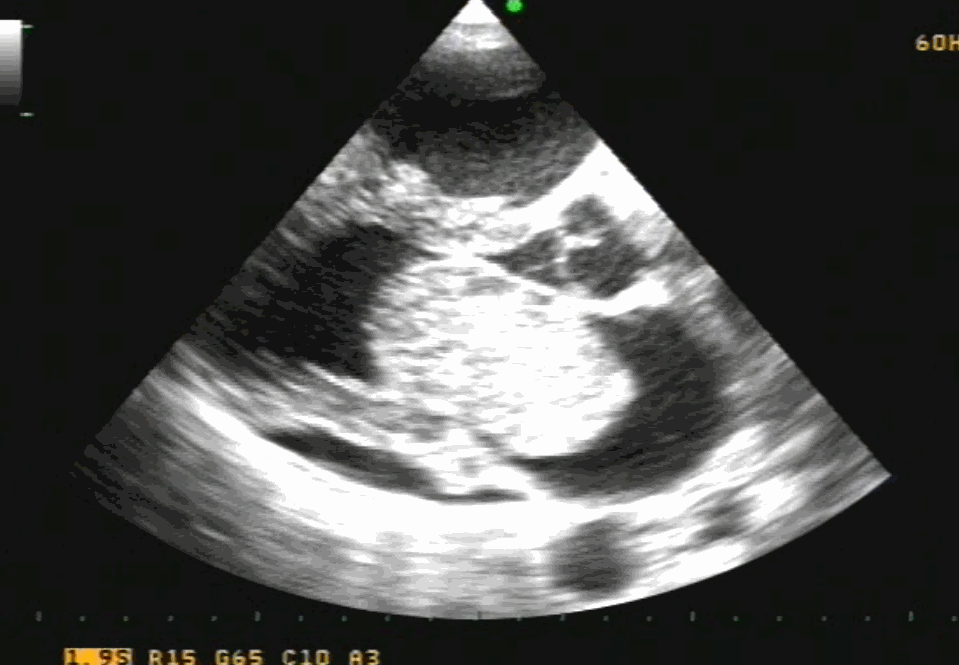
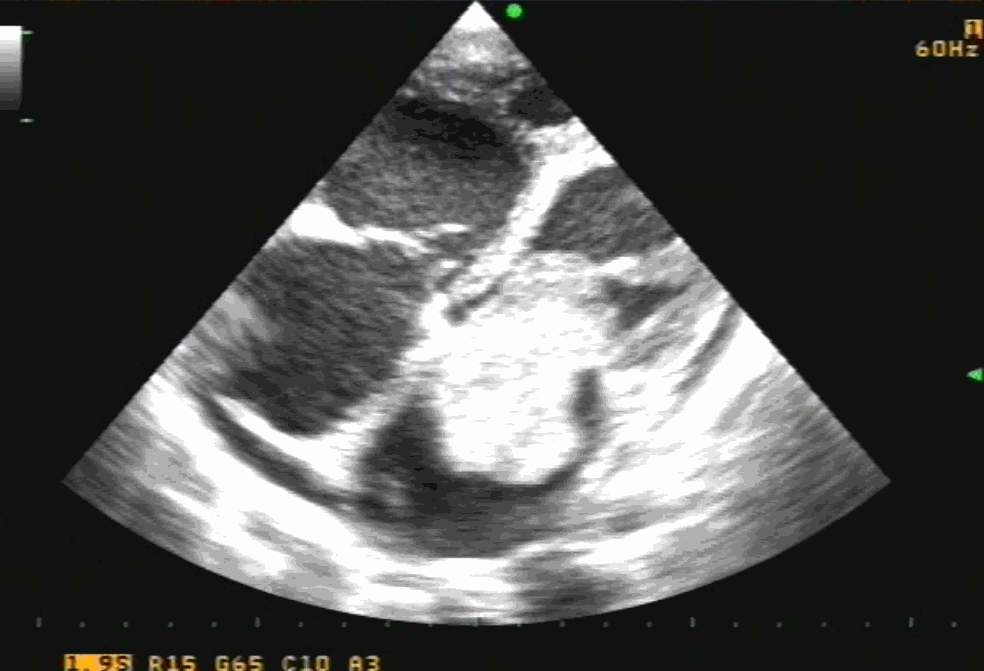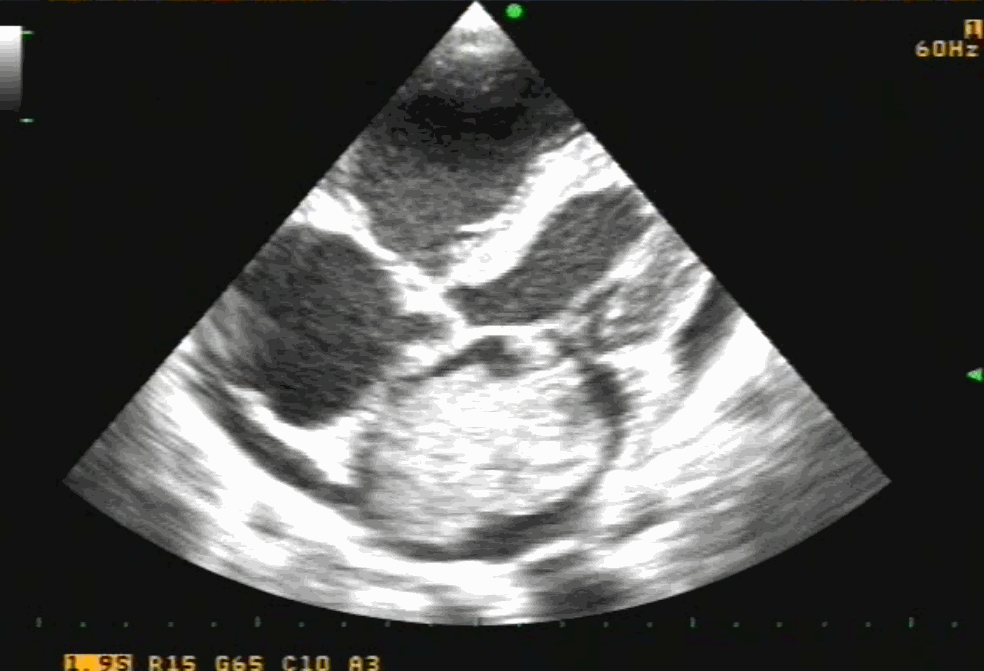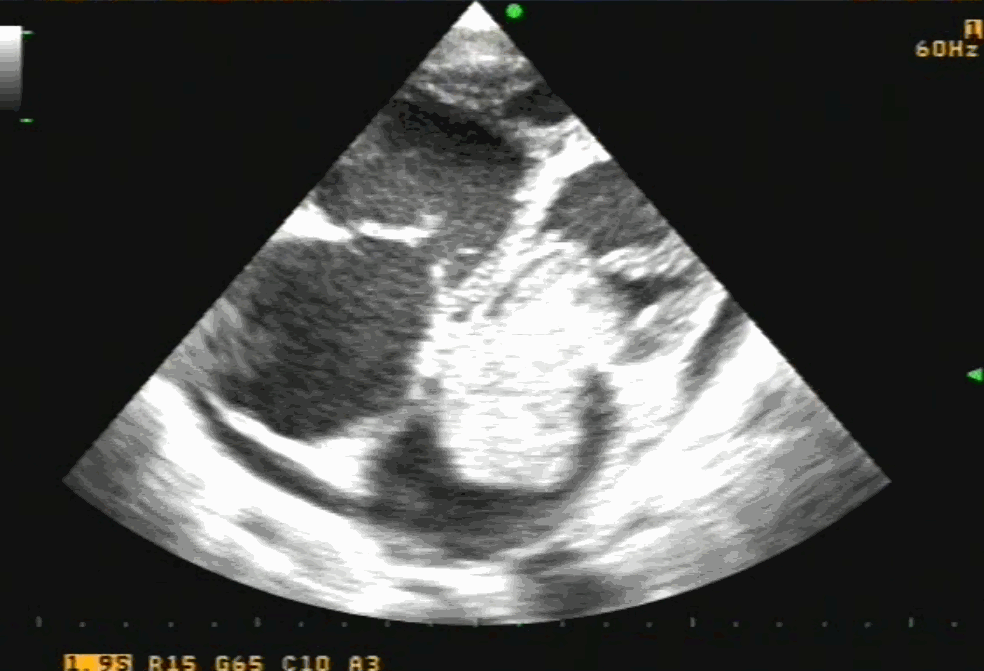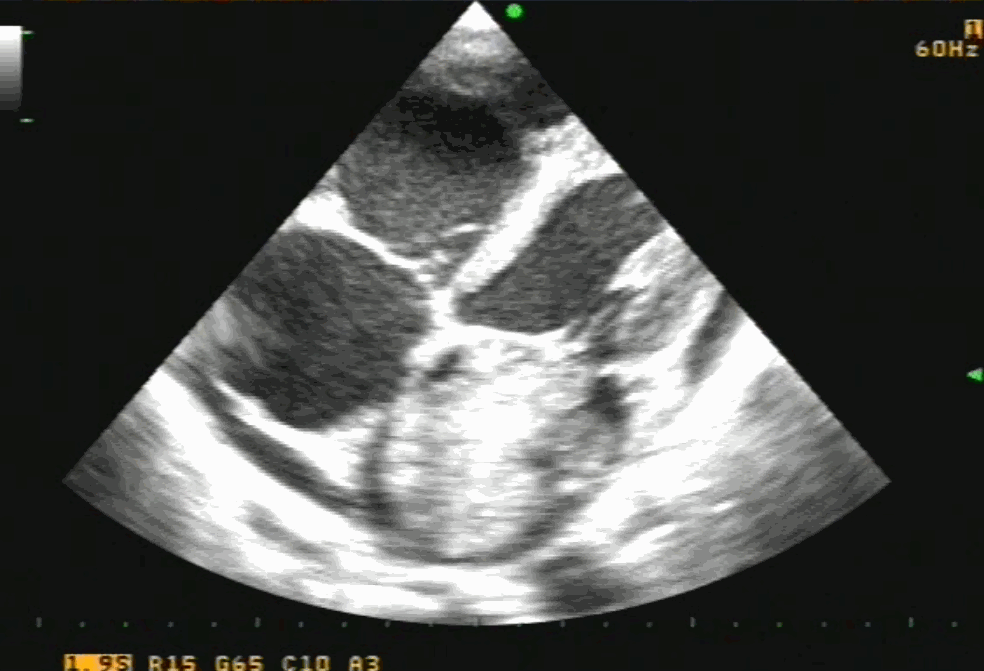
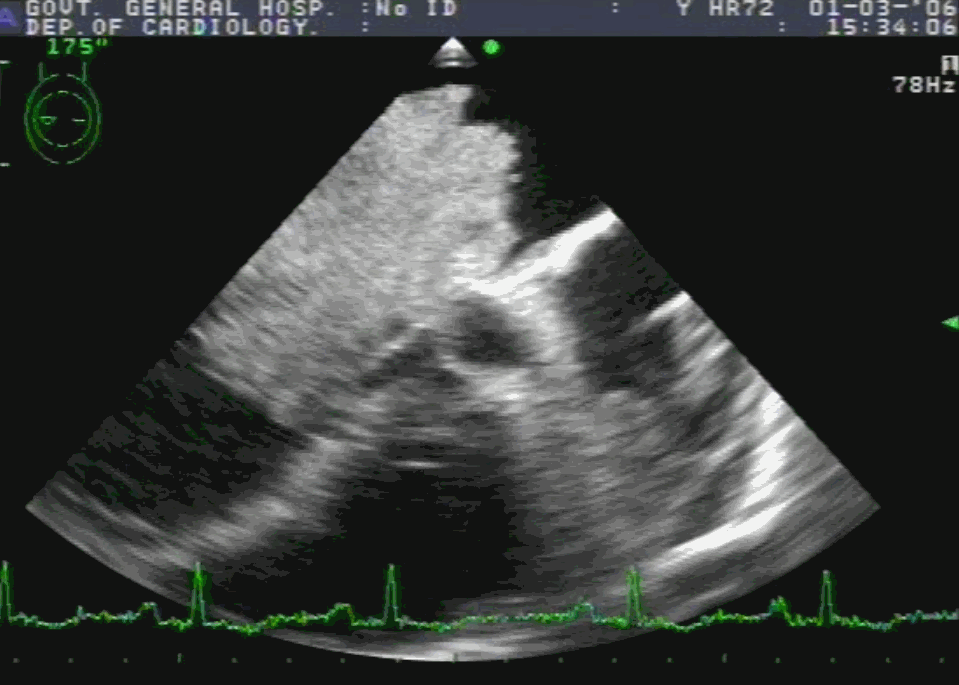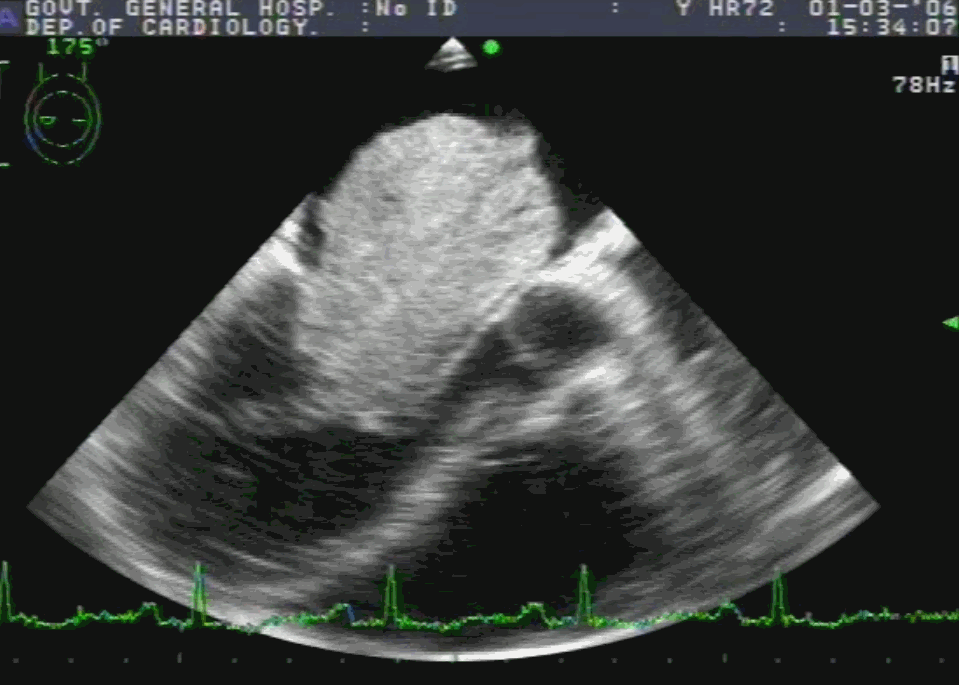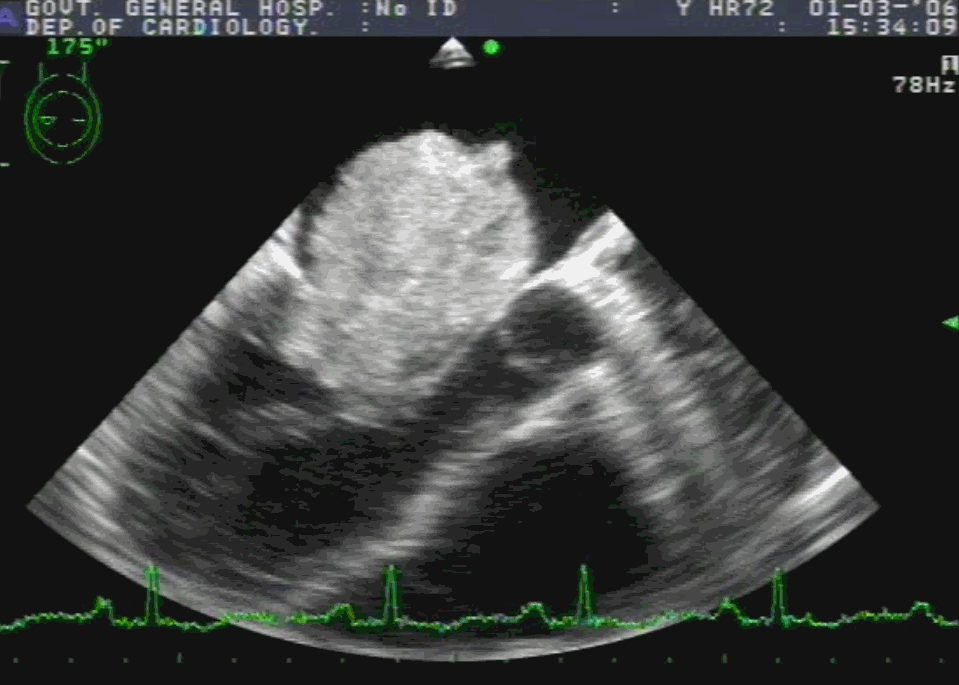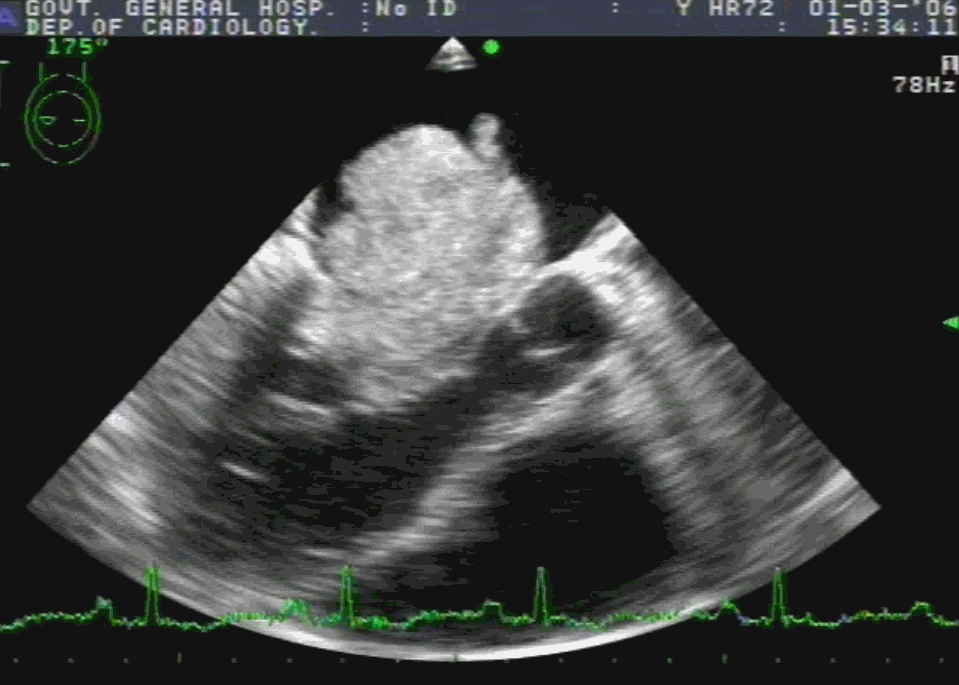
Sinus venosus ASD (also referred to as SVC ASD) is a defect in the failure of the sinoatrial orifice to lateralize completely to the right side during atrial septation.Left venous valve, as well as the septum secundum, fails to fuse with the roof of the atria creating interatrial communication. During this process, the developing pulmonary vein overshoot to the right side making PAPVD a mandatory add-on defect. (Harley ,Thorax 1958 ) It can be referred to as embryonal venous migration defect at the level of RA. In the same sense, it is not a true defect in IAS but a defect in septation between SVC/PV. It may also be referred to as unroofing of RUPV. The so-called Inter atrial communication actually is the confluence point of RUPV/SVC/RA.(See TEE images below)

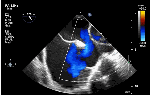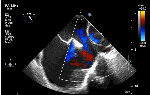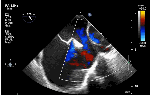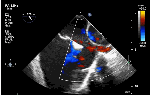
We know, in SVC ASD-commonest associated anomaly is PAPVC . It is not an ideal term to use though, instead, it is encouraged to use the term PAPVD (drainage) . Technically true PAPVC can not be connected to RA cavity as PV can connect either to cardinal or vitelline vein only. This distinction is helpful when we search for additional PAPVCs cranial to SVC. Sometimes we might recognize this error only after closure of SVC ASD.
Should we close SVC ASD ? How do we close?
Age, natural history, symptoms, the quantum of shunt will answer an occasional troubling query , “should we close it at all? Surgery is the standard approach till now. What makes device closure popular? Two reasons 1.Patient /or parent’s fear of surgery 2.Cardiologist’s urge for innovative Interventional procedures. (The fact that a simple covered stent will do the job is too tempting to make an attempt) However,please note, the procedure is not at all simple as one would Imagine.
Anatomical prerequisite for device closure
The defect must fulfill some critical anatomical essentials.
- It should be an isolated defect.
- RA should not be grossly enlarged
- Re-routing of RUPV to LA should be possible
- A significant circumference of RUPV should be committed to LA.
- There should not be downward extension involving septum secundum making it an SVC + OS ASD
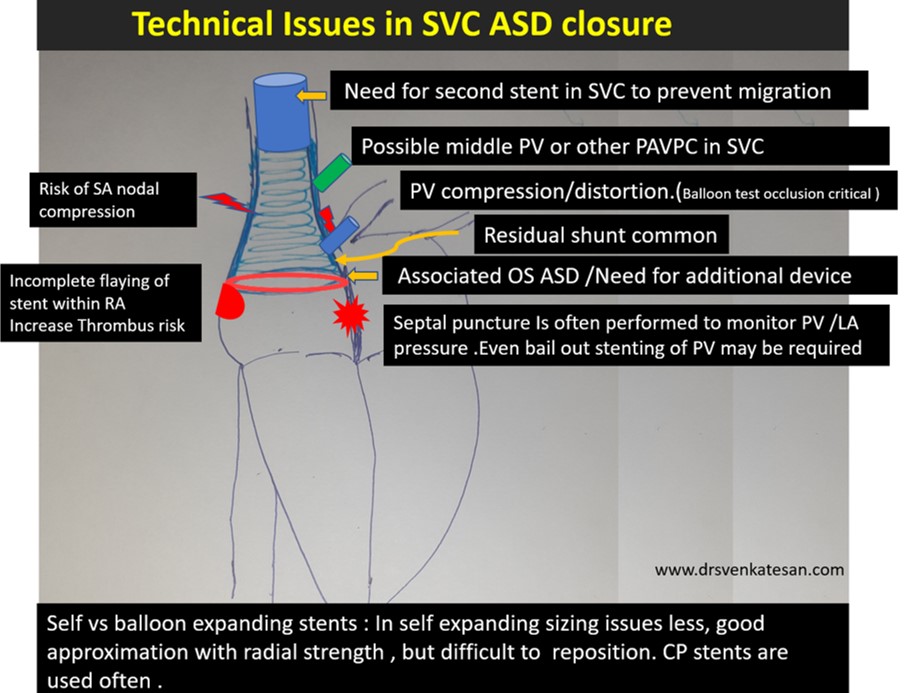
Technical issues
- Self-expanding vs balloon expanding stent (Anyone may be chosen)
- Stent flare-up SVC RA junction crucial
- Must ensure RUPV doesn’t get compressed with device.
- Forces that hold the SVC end of the stent is very important. Sometimes It may require a second proximal stent just to prevent migration of the first stent.
- Live LA pressure and RUPV monitoring may be critical to recognize PV ostial compromise. For this, a transeptal puncture may be required (Ironically creating another mini ASD !)
Finally, and most importantly follow-up is mandatory with device closure since the stent is on the venous circuit as RA, SVC thrombosis expected. (Anticoagulation protocol not clearly defined as of now )
Final message
Device closure for SVC ASD is a good Innovation. A perfectly delivered covered stent at the RA/SVC junction will do the trick. However, In my opinion, surgeons do a neat(More complete) job It is time-tested. Single or double patch or warden procedure may be done.(Ref 2)
Reference
https://doi.org/10.1016/j.pedneo.2019.06.013