Exertional dyspnea disproportional to the effort is the most common (cardinal)symptom of heart disease. Whenever we discuss the mechanism of cardiac dyspnea , we primarily attribute it to left heart disease, elevated LVEDP and the resultant pulmonary congestion.Conventional teaching in the past (may be in the present too !) doesn’t implicate raised RVEDP in the genesis of dyspnea.
It’s good to recall , the sensation of dyspnea is felt at the peri -Amygdala nuclear zone after complex processing with various cortical and sub-cortical level .It is subjected to as many afferent triggers other than J receptors in pulmonary micro circulation. (Eg Exercising skeletal muscle). It is believed, mechanical stretch receptors exist within the walls of heart along the sub-endocardial aspects of chamber.
(Muscle spindles which are the sensors of muscle tension are extensively noted in skeletal muscle that contribute to the origin of dyspnea .We are not yet accruing enough evidence whether cardiac muscle do have the same muscle spindle or it’s equivalents to cause dyspnea when stretched. However, we clearly witness in the practice of clinical cardiology , isolated elevation of RVEDP ( also RVSP ) to cause significant dyspnea in specific clinical situations.
Potential causes for Isolated Right ventricular dyspnea
- Pulmonary hypertension (COPD included* where in it could be a combination of lung and cardiac dyspnea)
- Acute pulmonary embolism
- RV Infarction
- Acute rupture of sinus of Valsalva aneurysm (RSOV) Here RVEDP is often > LVEDP and dyspnea is due to the acute stretch of RV
- Isolated normal pressure TR(RVEDP is low still cause dyspnea due to volume related RV triggers)
- Any RVOT obstruction (Classically valvular pulmonary stenosis)
- Does RV dilatation without elevated RVEDP cause dyspnea ? Though right ventricle is developmentally and hemo-dynamically better suited to handle volume , still, it struggles to manage sudden increase in volume .(Another clinical example is seen in patients who are on dialysis)
*RV diastolic dysfunction is still a Infantile hemo-dynamic concept .Whether it can raise RVEDP significantly during exercise and Independently contribute to dyspnea is at best a hypo-science.
Role of muscle spindle and mechno-receptors
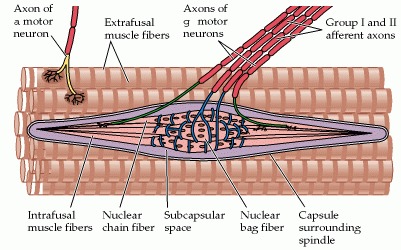
structure of skeletal muscle spindle. Though we don’t have a highly developed spindles in smooth muscle and cardiac muscle we have evidence to suggest cardiac neural ending do have mechano-receptors with afferent connection through visceral neural plexus that can trigger both heart rate and respiratory centers Further reading : Neuroscience. 2nd edition. Show details Purves D, Augustine GJ, Fitzpatrick D, et al., editors. Sunderland (MA): Sinauer Associates; 2001.
Bain-Bridge reflex: The hidden link in right heart dyspnea
Bain-Bridge reflex is a 100 year old concept. still helping us to understand the basics of right heart hemodynamics and how adjustments with acute volume loading take place.He proposed that veno-atrial stretch receptors are located primarily in great veins as it enter ,right atrium (RV as well).
This gets activated through vagus and stimulates in brain-stem sympathetic system and increase the heart rate to handle the excess blood reaching the heart. How often we feel the symptom of palpitation whether due to this reflex ( when it is operating) is not really tested. But, what we can infer is , the surge in sympathetic tone perceived can be perceived as dyspnea.
*Clinical Relevance of the Bezold–Jarisch Reflex and its possible interactions with Bain Bridge reflex is a different topic.

It is interesting to note many of these reflexes cause hypo-tension, bradycardia and hypopnea (Even near Apnea.) The word dyspnea is surprisingly not used .It is highly plausible many of the unexplained dyspnea we see in otherwise healthy population is attributed to acute or chronic volume overloading or under-loading of right heart.
Role of PFO in right heart dyspnea
PFO is a natural decompressing orifice in the IAS guarded by a flip-flap safety valve which is a remnant of septum primum .Though it can flow either way , since the flap of the valve is larger in LA side, it gets closed when LA pressure raises but opens up , if RA pressure raises making it more often a right to left shunt at times of elevated RA mean pressure. In isolated right heat pathology , this communication shunts right to left and adds a new dimension to cardiac dyspnea (Now, It becomes a hypoxic /biochemical dyspnea over and above the right heart stretch related dyspnea )
Other mechanisms in right heart dyspnea
Pulmonary arterial stretch and altered QP : Role of ventilation perfusion mismatch should also be considered as a cause for dyspnea in isolated RV pathology. The term V/Q mismatch is a poorly understood term fro me. My Inference is, since RV contraction provides the Q in the equation V/Q .Whenever Q falls V has to fall to maintain neutrality causing net hypoxia and dyspnea.
Final message
Dear fellows, never hesitate to attribute the origin of dyspnea, to elevated RA mean pressure /RVEDP. It is due to RA/RV stretch secondary to volume and pressure overloading with a perfectly normal pulmonary capillary wedge pressure or LVEDP. As in the left heart ,this occurs both in pathological as well as perfectly exaggerated physiological times.
Reference
1.Bainbridge FA. The influence of venous filling upon the rate of the heart. J Physiol. 1915 Dec 24;50(2):65–84. [PMC free article] [PubMed] [Google Scholar]
2..A J Crisp, R Hainsworth, and S M Tutt The absence of cardiovascular and respiratory responses to changes in right ventricular pressure in anaesthetized dogs. J Physiol. 1988 Dec; 407: 1–13(This paper actually undermines the importance of RV receptors. It is still perplexing to note both the inflow into RV (ie RA and the out flow pulmonary artery circuit has richly innervated by receptors , its difficult to accept why we have failed to get much evidence for RV stretch receptors) Its potentially great area of research for cardiac physiologists. That will be a tribute to the greats like Bain Bridge and Bazolds Jarich.)







