Every day, 100s of physicians and cardiologists are asked to report ECG either physically or on-line .In India, they do it for some paltry benefits from stand alone labs , hospitals , institutional protocol or by sheer compulsion.
When only ECG is reported ,the physicians are often blind to the indications for which it is taken . The symptoms of the patient or the past history is rarely available. It is a sorry state of affairs and many of us do this, fully aware of the consequences – a normal ECG can’t rule out an ACS in at least 10 % of times.
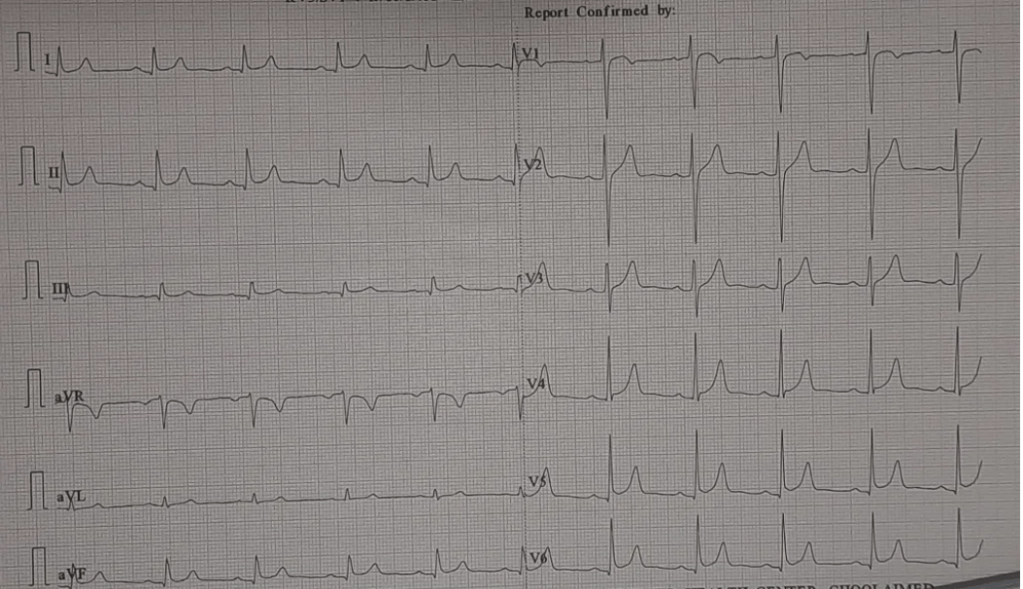
Now, here is an ECG of a 40 year old male, on my table for reporting. No other info is available.
How should I report it ? Can I refuse to report ?
There were five options
A.ECG Normal
B.ECG within normal limits, with non specific ST/T changes
C.ECG shows ST elevation Infero- lateral leads. To rule out ACS
D. Early repolarization pattern
E. I refuse to report, until clinical data is made available.
My report: ECG within normal limits. (Shows ERS pattern with non specific ST elevation in Infero-lateral leads.To correlate clinically, and advised a physician consult. )
One of my colleagues commented, that I lacked the guts to comment the above ECG as normal. The contention is, ERS pattern occurs in up to 20% of the young population, and it evokes unnecessary anxiety. How do I know he is pain free ?However, I agreed with my colleague. It is indeed ERS by all means. But,how long ?, we can be carriers of proxy anxiety, for the sake of our patients. Further, by no means ERS gives immunity to ACS. (What happens to the ST segment of ERS during ACS is a different query to be answered.)
(*Later on I came to know this guy had epigastric pain, and was simultaneously evaluated for CAD which was excluded . Mind you, mistaking ERS pattern for ACS is an age old problem. About 5% of thrombolysis in ISIS -2 study(1988) turned out to be in patients with possible ERS)
Final message
Can we report an ECG without knowing the patient and his symptoms ?
It is a foundational question in clinical medicine. The answer to which can shake the way, we practice cardiology. It is indeed unprofessional* of a physician to report an ECG without knowing the patient status. But we do it every day , can’t avoid it. But ,let us at-least insist the ECG lab guys to note down the symptoms or /the purpose for which it is taken (like a regular health check or pre-op evaluation, etc).
*Of course , to escape from a potential miss, we should atleast write, ECG to be correlated clinically and with past records.
Post-amble
We are in the AI era. ECGs are read by data-hungry machines. If you think there are AI models that can match a million ECG patterns and detect an ACS in milliseconds, sorry, you are sadly mistaken. Unless and until patients’ entire history is fed into humanized machines, (that had undergone 8 years of rigorous cardiology training) the risk of missing a diagnosis is significant.