Atrial septal defects are one of the commonest forms of congenital heart disease.
- The commonest being the ositum secundum ASD ( Which is in fact is a defect in the development of septum primum)
- The next common is ostium primum defect which is a part of AV canal or atrio ventricular septal defect.
Other forms of ASD include
- SVC type /Also called sinus venosus type of ASD .
- IVC type
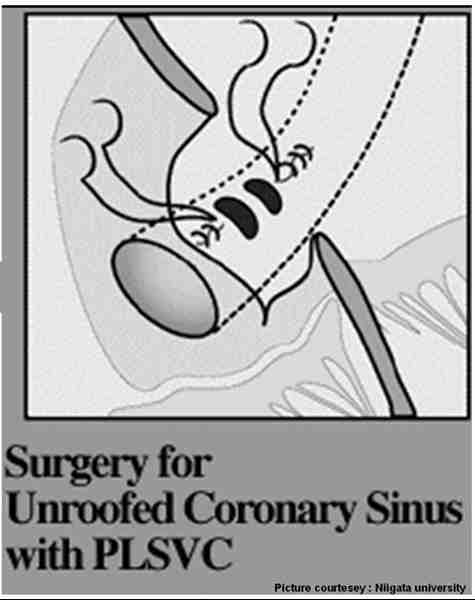
- Coronary sinus defect -Also called partial or complete forms of unroofed coronary sinus

SVC type ASDs
They are in the strict sense can not be called as ASD. This is because there is no defect in any of the embryological inter atrial septal component.
There is no direct communication between RA and LA, instead a window or passage of communication between pulmonary vein and SVC. Right upper lobe pulmonary vein is usually the culprit .Some times more than one PV communicates with SVC.
The exact area of this PV-SVC window occur between anterior surface of right upper lobe PV with postero lateral surface of SVC.
PAPVC partial anomalous pulmonary venous drainage can be considered an integral part of this defect as RUPV is linked with SVC.
Can we have a combination of SVC ASD and OS ASD ?
This is possible .But two embryological errors need to occur. This is often seen as a large OS ASD with deficient or absent superior rim. So whenever superior rim of IAS is deficient a PAPVC and a SVC ASD should be looked for.
Clinical features
- SVC type ASDs generally shunt lesser blood than OS ASDs. (Often<2:1) .This is because it is not the LA that is communicating with RA instead only a
- It is usually a single PV (some times 2) that shunts from left to right.
There is a distinct possibility of missing this lesion in routine echo.Minimal RA,RV enlargement may give us a clue.The classical subcostal or 4 chamber view in echocardiography may not visualise these defects.
So, whenever one encounters mild dilatation of RA and RV and the IAS appears intact, a meticulous search and a focused echo in the superior aspect of IAS is warranted. Angled superior views may pick up this defect.A transesophageal echocardiogram (TEE) is often required to confirm it.
Therapeutic issues
- Device closure is not possible
- Surgery involves little more technicality than ASD OS.
- Small defects can be patch closed.
- Some times the SVC has to be disconnected from the PV and anastamosed separately on right atrial appendage. SVC resection will aid the surgeon in proper patch closure.
- Post operative follow up is necessary as SVC obstruction or PV obstruction may be a delayed consequence
References

http://ats.ctsnetjournals.org/cgi/content/full/59/6/1588?ijkey=4f42649bc5805f3cf7d60f5728ec7f871356277b
http://asianannals.ctsnetjournals.org/cgi/content/full/10/3/231
Read Full Post »