Archive for the ‘Uncategorized’ Category
A forbidden quote in medical science
Posted in Uncategorized, tagged empircal medicine, ethics in cardiology, evidence based cardiology, evidence based medicine, FAKE VS TRUE EVIDENCE, jama network, lack evidence, lancet, MBEST QUOTES IN MEDICAL ETHICS, MEDICALEDUCATION, nejm, randomised controll trials, venkatesan sangareddi on September 14, 2024|
Can we differentiate “Sarcomeric vs non Sarcomeric HCM” by Echocardiography ?
Posted in Uncategorized on September 4, 2024|
Echocardiography, the ultrasonic vision, with which we are able to directly visualize the heart is a monumental discovery ,gifted to us by Edler & Hertz in the 1950s. These high frequency sound interacts with myocytes in a variety of ways like penetration ,reflection, ab & adsorption, back scattering etc. It has further evolved , at the tissue level diagnostic like 3D speckle ,and strain etc .
With all all these advancements ,how good is echocardiography in ruling out (or in) reversible /treatable HCM mimickers or their molecular subsets.
Recognizing non-sarcomeric Hypertrophic Cardiomyopathy (HCM) by echocardiography is one such task .It can be challenging, but here are some clues to help:
- Unusual hypertrophy patterns: Non-sarcomeric HCM may exhibit atypical hypertrophy patterns, such as:
- Mid-ventricular hypertrophy
- Apical hypertrophy
- Hypertrophy in the right ventricle
- Absence of systolic anterior motion (SAM): Unlike sarcomeric HCM, non-sarcomeric HCM may not exhibit SAM of the mitral valve.
- Normal or reduced left ventricular outflow tract (LVOT) gradient: Non-sarcomeric HCM may not have a significant LVOT obstruction.
- Presence of fibrosis or scar tissue: Look for signs of fibrosis or scar tissue on echocardiography, such as:
- Bright or patchy appearance in the myocardium
- Increased echogenicity
- Abnormal myocardial texture: Non-sarcomeric HCM may exhibit an abnormal myocardial texture, such as:
- Speckled or “ground-glass” appearance
- Other structural abnormalities: Some non-sarcomeric HCM cases may exhibit other structural abnormalities, such as:
- Mitral valve abnormalities
- Tricuspid valve abnormalities
- Right ventricular abnormalities

Ref : Sarcomeric versus Non-Sarcomeric HCM June 2023 Cardiogenetics 13(2):92-105
Some specific echocardiographic features of non-sarcomeric HCM include:
- Fabry disease: Typically shows a “binary” appearance of the myocardium, with a bright and dark pattern.
- Amyloidosis: Often exhibits a “granular sparkling” appearance of the myocardium.
- Cardiac sarcoidosis: May show a “starry sky” appearance due to myocardial fibrosis.
Please keep in mind, that echocardiography should be complemented with clinical evaluation, genetic testing, and other diagnostic tools to confirm the diagnosis.
Amyloid -HCM coexistence
Medical pathology is never a pure science.It can make our assessment topsy turvy at any moment .One such rare phenomenon is amyloid getting deposited in a patient with classical inherited HOCM. (Boyangzi Li, Cardiac AA amyloidosis in a patient with obstructive hypertrophic cardiomyopathy,
Cardiovascular Pathology, Volume 48, 2020,)
A comment on deep genetic profiling
Genetic studies in HOCM are academically exciting and professionally gratifying. From the patient perspective, it provides an opportunity to treat any reversible or treatable enzyme disorders(Ref 1 Migalastat for Fabry’s disease & Tifamidis and Patisiran in TTR Amyloidosis) .
But, the benefits of deep genetic testing has to be carefully harvested as there is a troubling trade off, due to the hitherto hidden prognostic anxiety, these genetic breakthroughs bring along.
Final message
Contrary to the belief , a carefully interpreted echocardiography along with clinical profile will be able to recognise sarcomeric HCM in most situations.But, it is still weak in ruling in a non- sarcomeric HCM.
Reference
2.The small interfering mRNA drug , is an interstitial protein excretory drug in TTR amyloid.

The pseudo-superiority of pPCI is exposed one more time … but it will thrive!
Posted in Uncategorized, tagged captim trial, prehospital thromolysis, primary pci vs prehospital thrombolysis, primary pci vs prehospital thromolysis, stream, stream 2 trial on September 1, 2024|
CAPTIM trial was published in 2002, which left a gospel truth in the science of myocardial reperfusion (two decades gone now). It is a sad academic story ,most of the interventional cardiology community shrugged it off as a non-event. After CAPTIM , there were several other studies that tried to reiterate the same. The fact of the matter is, in the art and science of reperfusion, all that glitters may not be gold at the myocardium level.
Yes, we do have a tiny advantage of pPCI in terms of complete revascularisation. But it was blown out of proportion, backed by small but glamorous studies. However, the true benefits are hidden in a timely early reperfusion, best done at out-of-hospital (or even in-hospital*) with the emergency crew’s assistance.
“Since Intravenous lysis looks too simplistic, that do not need expertise, and lacks a commercial trail, it is wrongly depicted as inferior management strategy in STEMI“
Every one of us is equally responsible for this sorry state of affairs. In many countries, the hub-and-spoke model is struggling to deliver. The spokes often fail in their duty to begin the reperfusion, (Perceived low quality treatment and peer pressure) while the further delay happens at the hub , that earn the wrath of the myocardium ultimately. In this context, we need a movement to revive the pre-hospital thrombolysis. This is what CAPTIM told us.
No one knows how the pPCI related delay was legally ratified and academically accepted by the elite cardiology forums. Some poorly designed small cohort RCTS are responsible for this. There were some sensible moves too later on. To counter the logistic limitation of pPCI came the pharmaco-invasive strategy backed upby STREAM, FAST-MI trials etc. Still, no one is able to undo the disproportionate popularity of pPCI .At best, It has a miniscule 1 % edge in the outcome if performed on time, at a expertise intensive place. (Efficiency of thrombolysis is highly reproducible. It is a fact, an ambulance crew is able to equal or even surpass the expertise of cardiologists in terms of absolute myocardial salvage) )
Now, it is heartening to read a meta-analysis addressing pre-hospital thrombolysis, done from my part of the country. This paper is published by Dr. R. Bharathkumar, and his team. He is one of our senior colleague, mentor and former professor of Stanley Medical College. Chennai. With this study ,they have successfully proved an existing truth, in a more refreshing and authoritative manner (Ref 1 )
I am here with sharing this full paper. Readers can infer their own conclusion. Would like to to highlight two important new points told in this paper in a gentle way, yet conveying a powerful message.
1. Much surprisingly, the guideline recommended the “door to needle time” in pPCI is achieved only in a fraction of the population in real world. (NRMI data Ref 2)
2.There is a differential Impact of PCI related delay on the myocardium with reference to IRA. In LAD STEMI time is more crucial. The permissible limit is 40mts not the traditional 90-120 minutes (Ref 3)

Final message
The perceived superiority of pPCI over any other modality of reperfusion is not absolute*. The above paper reemphasize this one more time ,with strong evidence aggregation .I wish ,this paper deserves a more prominent place in major journals like Lancet or JACC or even NEJM.
Meanwhile, we should not make sweeping statements, that tend to convey a wrong idea, which makes pPCI look invincible at all points in ACS time line. The irony is , many of us strongly believe, it is worth loosing the golden hour in lieu of perceived superiority pPCI . This shatters the concept of “time is muscle ” Thanks to the evidence based cardiology.
Counterpoint
*PCI in the setting of ACS do have a critical life saving role in certain subsets of ACS, that can never be undermined . What is being debated is the true benefits of this revolutionary intervention as a population level strategy.
Reference
What happens to PR Interval in Sinus bradycardia?
Posted in Uncategorized, tagged first degree av block in sinus bradycardia, pr interval in sinus bradycardia, sinus bradycardia, vagal tone in sa vs av node on September 1, 2024|
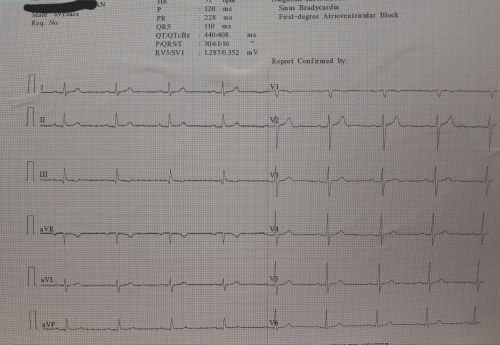
This ECG can help you find an answer .HR is 50/mt. Find the PR interval .It is also printed right there.
What happens to PR Interval in sinus Bradycardia ?
A. Remains normal
B. Prolonged but within normal
C. Prolonged beyond normal
D. Depends on the cause of Sinus bradycardia
Answer
When cardiac cycle slows down, every interval must get prolonged. PR interval is no exception. If you apply that rule the answer would be simple (Its B ).But this question has much more issues hidden into it. In this ECG it touches on the doors of first degree AV blcok. Normally PR doesn,t stretch that far in isolated benign sinus bradycardia.
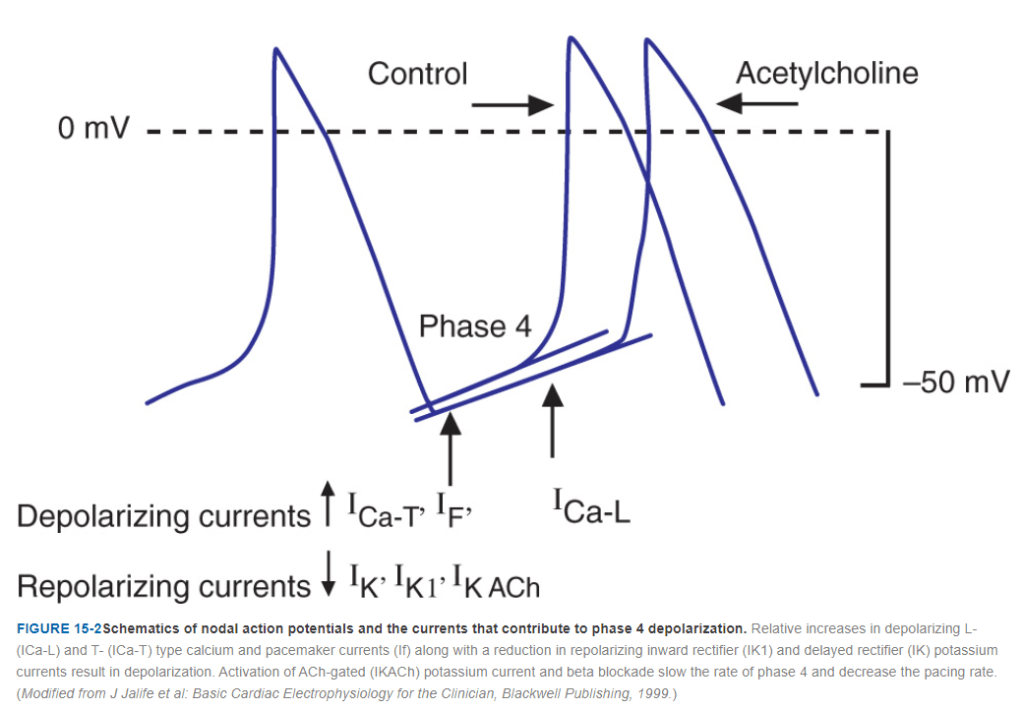
Sinus rate is determined by SA nodal, funny pacemaker current(if) .The rate of which is determined by the delicate balance between sympathetic and para-sympathetic signal flux. The dynamicity of the slope of phase 4 , ie spontaneous resting membrane is another key determinant apart from sift in the density of mean P cell firing focus .(see below)
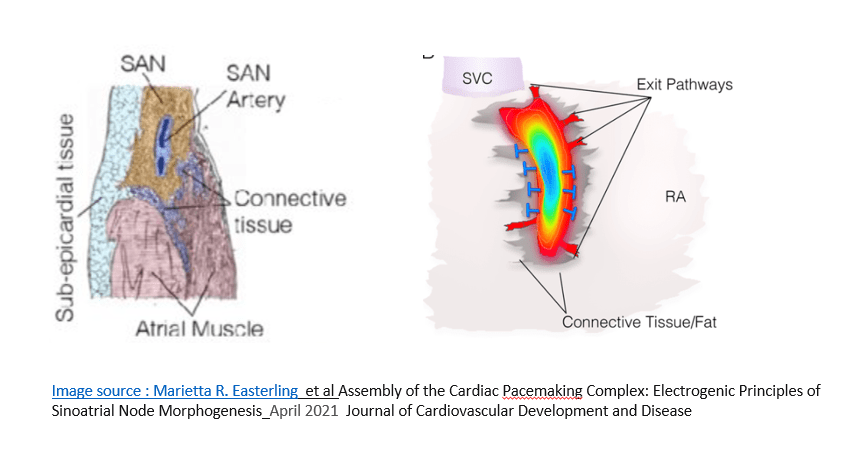
The sinus rate depends upon the cranio- caudal shift that occur within specialized tissue .(10-15mm slender the basmati rice shaped SA node)
When do PR interval prolong in sinus bradycardia ?
The commonest cause of sinus bradycardia is due to increased vagal tone . It is no secret , vagal tone do Influence AV node as much as it does the SA node . So, what can we expect ? Logically .the AV node must also slow down . But does it happen really ?
No, most of the time the AV nodal delay doesn’t not occur in sinus bradycardia . Does that mean the vagal spill over do not reach the AV node ? It does, but the fact is, the reduced heart rate attenuates the normally prevalent decremental conduction property of AV node. So we do not expect a prolonged PR in sinus bradycardia.
However, if PR interval is prolonged in sinus bradycardia , we can except silent pathological states like , sinus node dysfunction where AV node can also be affected. Also, prolongation of PR is very common in ACS situations in Infero- posterior (RCA/LCX) territory where both SA and AV nodes are simultaneously targeted.
Prolonged PR in bradycardia some times is a hemodynamic necessity as ventricular filling time is prolonged.
Clinical Implication
It is a good habit to have rapid glance at PR interval in every patient with sinus bradycardia. This will ensure a rare miss of AV nodal dysfunction .
Final message
PR interval is normal or show little prolongation in most of sinus bradycardia. If it is prolonged, without any circumstantial evidence for enhanced vagotonia , then, it could indicate some thing wrong in the conduction system. Atropine test might help us out in differentiating true vagotonia from intrinsic delay.
Why many Ischemic VTs are not abolished by revascularization ?
Posted in Uncategorized on August 19, 2024|
Who said this non-sensible statement? ( I guess ,this would be the first response from many cardiologists !) If you feel the same, then this post might not be for you.
Relationship between Ischemia and arrhythmia
While the relationship between Ischemia and VT is really complex, the term “Ischemic VT” itself adds more twists. Its all about timing, intensity of Ischemia , associated factors and finally the baseline arrhythmic risk that includes the mystery defects in myocyte gap junctions and ion channels.
Following are some of the observations.
Primary VT
This is the true Ischemic VT. Even here, it is the associated factors, like hypoxia or acidosis are the triggers which of course are resultant of Ischemia. There are further problems . Even critical Ischemia, as in high grade unstable angina, rarely Initiate a VT, while STEMI seems to have the exclusive rights to trigger it , by its ability to produce acute transmural ischemia . (Note: Whether primary VT occurs before or after myocardial necrosis is not clear) There is evidence to prove, susceptibility to VT at times of Ischemia is in the genetic make-up of ion channels, as pointed out by famous French electrophysiologist Haïssaguerre. (NEJM 2008)
Post Infarct VT : (24 hrs to 2 weeks ,an empirical criteria we use)
This can be called as Ischemic, still we ‘wouldn’t know whether the arrhythmia is originating from dead or live tissue. It can even be combined Ischemic-Scar VT

Late Ischemic VT
These are the typical scar – substrate -mediated ,micro/macro reentry VT .The strip of tissue on the border zones of conflict (between viability and non viability is always restless (Gaza strip of VT?) This is rarely amenable to revascularization, unless some one is able make that area 0% Ischemic , which is a highly improbable scenario. The alternate option is diagonally opposite .EP guys are empowered with a deadly solution, and authorized to shoot down focus (or isolate) instead of the futility of revascularisation. (Please note, this doesn’t work and should not be attempted in early ischemic VT, though few case reports of RF ablations during VT storm li- Juan Qu et al AMJS 2924 )
Final message
The relationship between Ischemia and VT is poorly understood, (rather than to say complex.) It is true ,acute Ischemic VTs has more closer relation with Ischemia, often settles down with prompt revascularization.
In chronic VT , shooting down the ischemic focus by ablation is more likely to extinguish the arrhythmia ,rather than revascularization. This is because partial revascularization irritates the viable myocardium and keep the ischemic focus active. ( Class C evidence) ICD though a revolutionary technology to prevent a SCD in these circumstance it makes a poor choice to reduce the arrhythmic burden .At best , it is just a back up device to tackle the escaped VTs in spite of RF ablation and drugs.
Reference
Its time WHO,may declare CVD as a “communicable disease” equivalent
Posted in Uncategorized, tagged bmj, cvd prevention, jama network, journal of medical ethics, lancet, medical education, medicalethics, nejm, prevention quotes, sdg, sustainable development goals, who on August 5, 2024|

*Lifestyle definition
A set of attitudes, habits, or possessions associated with a particular person or group. and such attitudes, etc, are regarded as fashionable or desirable.

Final message
Communicable disease need not be an Infectious disease like covid. The word “Communicable” shall soon convey a new meaning, to the enlightened. Adverse life styles ,disseminated into the community that vigorously propagate CVD, has every reason to be referred to as a ‘Neo non-infectious pandemic”
Postamble
In the strict sense, CVD is not a communicable disease ,rather the risk factors are …but technically it is.
Reference
3.A comprehensive narrative review

Trying to answer two casual queries on RV dysfunction
Posted in Uncategorized, tagged lv function impact on rv function, rv dilatation vs hypertrophy, rv dysfunction, tapse limitations, transverse vs longitudinal rv function on August 3, 2024|
1.What is the response of RV to pressure overload ?
A. Dilatation
B. Hypertrophy
C Both occur together
D. Hypertrophy is the Initial response, followed by dilatation
Answer :
Since we believe RV’s behavior is generally opposite to that of LV , many would tick, dilatation as the first response. This may be correct when there is acute raise in RV after load, as in PE. However, It is surprising even in chronic pulmonary hypertension , the degree of RVH is not constant and homogenous .This is because , different parts of RV chamber has different wall thickness .Further, the pressure distribution from PA to RV is uneven. The co-existing TR confounds the loading conditions. It is not yet clear, how the RV would respond to raised PA pressure. In the bed side, we are seeing both flight(dilate) ot fight (RVH) reactions from RV (more often the former than the later) It is possible RV behavior is be pre-programed and built into the genes of the contractile proteins.
It is worthwhile to note, RVH is constant feature in non pulmonary hypertension related “after-load” conditions as in valvular or sub valvular PS. This is more to do failure of regression of RV mass early after birth, rather than the actual effect of high after load. Another point is purely technical. RVH is measured in RV free wall, in subcostal view in diastole and inspiratory phase.(upper limit is 4mm) Many of us could miss RVH in routine echocardiography unless specifically looked for.
2.Which is the first echocardiographic parameter to get impaired when RV fails ?
A. RV FAC (Fractional area change)
B.TAPSE
C. RV Ejection fraction
D.RV longitudinal strain
E. RV S‘
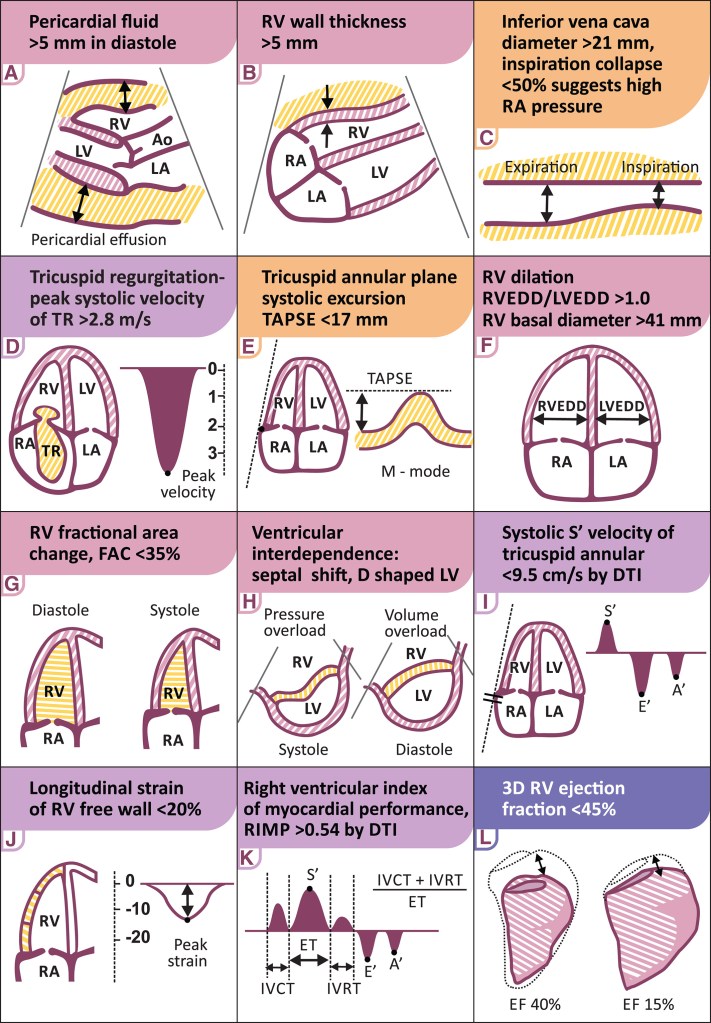
Answer : I am not very sure about the right answer , but TAPSE is last to get Impaired .(Still, we celebrate it like anything is a different story) Many believe transverse functional Indices like FAC is impaired early. and is less influenzed by the spurious spill over of Left ventricular contractile force in transannular plane (Which augments longitudinal functional Index like TAPSE),
The following illustration (From Ref 2 ) summarizes all RV functional parameter in a succinct fashion. Fellows must be familiar with at-least half of them.( RIMP is less practical and error prone can be ignored)
Reference
Aortic dissection reporting format: Not complicated, yet comprehensive.
Posted in Uncategorized on August 2, 2024|
Though foundational and prodigious, moving beyond De-bakey ,Stanford is a necessity in the management of Aortic dissections in current times. The first step is to understand the perfect 3 dimensional anatomy of entire Aorta from its origin to bifurcation and even beyond. (The 11 or12 segment demarcation of aorta is well established and gained acceptance.)

The society of vascular surgery and society for thoracic surgery has come out with land mark nomenclature in 2020.
This labeling tells us instantly about whether the dissection is A, B, or Indeterminate .It also reveal the origin of dissection, extent of dissection and the exit point, if available.

Naming & coding of Aortic dissection


Aortic dissection : Management cues
The above scheme is just a part of Initial work up with the help of MR angiogram or spiral CT. There are more critical factors like, clinical stability, time since dissection, false vs true lumen identification, its volume ,rate of propagation, branch involvement, mal-perfusion , etc need to be counted.
The curious Irony about this dreaded entity lies in the fact that in type A dissection, the surgical team need to be alerted well before they embark on this most complex cardiovascular emergency, usually in a state of the art CTVS unit. Meanwhile, most uncomplicated type B dissection demands total inactivity on the part of surgeon(as well as some cardiologists !) while the patients can be casually shifted to the intensive care ward, essentially for monitoring , bed rest and few drugs to reduce BP and shearing stress on aortic wall. The dissection can heal themselves.
The role of Interventional cardiology in Aortic dissection is evolving rapidly , still at best supportive or can buy time to bridge to surgery in some late presenting type A dissection. There has been lot of experience in some centers, where type B dissections are exclusively managed by scaffolding. But, the concern is, catheter based Interventions in low risk subsets of dissection is always a tricky decision.
Medical strategies should never be looked down upon as enemy of endovascular Interventions. So, one of the live and debatable issue is, how & where can we fit-in the hyper-talented endovascular Interventionists in the complex vascular arena of aortic dissection.
Reference
Two troubling medical quotes in tandem …
Posted in Uncategorized, tagged doctor patient relationship, medical ethics, patient demands, patient democracy, patient empowerment, patient rights and duties on July 28, 2024|
This one, is not an easy question about VPDs !
Posted in Uncategorized on July 19, 2024|
A 40 year old women with palpitation found to have complex multiple VPDs and elevated thyroid hormones.
The GP has refered for further management to a cardiologist, frightened by the morphology and frequency of VPDs.
The cardiologist has sent him to a EP guy .I guess he was not briefed well about the patient, and he decided to do , what he is best at .He tried to fix and shoot down the VPDs. The apparemt inappropriate procedure went on, as per the demand of patients and science . Now, let us question the basics .
The question is,
Where will be the focus of VPD in hyperthyroidism?
A. LV apex or septum
B. RVOT
C.Papillary muslce of LV or Intra -cavity
D. It is an invisble microrentry , or automaticity. Focus can be anywhere and can not be loclaised.
E. Technically there can not be a focus, it is simply enhanced adrenergic drive by free T 3 & T 4
Answer
There has to be a focus for every arrhythmia.It is my thinking. Some of my EP colleagues, say once circuit is established the focus looses it value . In systemic causes of cardiac arrhythmia , there need not have a visible focus. Make a Pardon , as of now , I can only frame a question, not the answer.
Final message.
Leave alone the answer to this question, I am sure every physician knows the correct treatment. Treat the cause, forget the manifestation. If some one is adamant he can do a RF ablation …not in the heart , but in thyroid gland.
Postamble
Even though , it is hyperthyroidism, we have a responsibility to rule out any tissue level substrate because, not every hyperthyroid patient throws this much of VPDs. It is highly possible, thyroid hormomes can un-mask a hitherto non -arryhthmogenic myocardial focus.