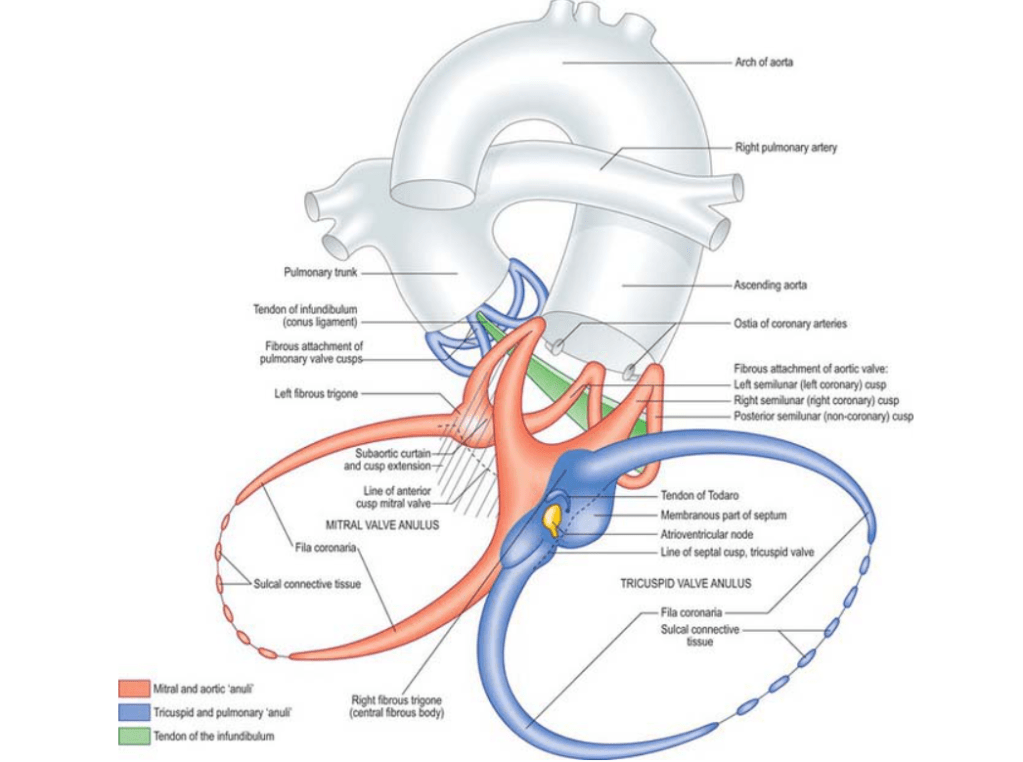
It is not at all a secret, among the four heart chambers, right atrium receives the least attention from cardiologists, as it is perceived merely as a conduit for venous return. (Why we forget, the fact? that the same blood that is handled by RA, would become the left ventricular stroke volume few beats later) .
Ironically ,”a” wave in the neck is celebrated for over a century as a evidence for a brisk and forceful contraction of RA. Meanwhile we also observe, with the onset of atrial fibrillation (AF) there is no dramatic hemodynamic deterioration in most people, reinforcing our assumptions might be correct after-all ! (Which is not)
So, we must admit , we have some difficulty to understand the true power of right atrium. But, as now, as on 2026, we begin to realize RA’s role is central in filling the RV, and thereupon the Pulmonary circuit and LA. It is also not clear, why RV frank starling law is rarely considered important, in spite of the fact, RV wall stretch is directly dependent on RA volume.
What is normal RA volume ?
(See below) Technically every chamber must have stroke volume plus something , that will remain as residual after its emptying. In that sense, RA volume is about 100 ml. (During exertion, it can go even to 150 ml). One curious issue in the morphometrics of RA is, unlike the other chambers, the inlet boundary for RA is vague as IVC and SVC simply blends to form a tubular chamber without any valves. During peak exercise, one may not distinguish the physiological limits of the inlet of the right atrium.
Table taken from Ref 1 . Note : There is something called RA ejection fraction. Might be of immense importance in right heart failure.
What happens to RA volume in cardiac failure ?
It is no surprise, the thin-walled RA enlarges more than RV. Also, in the chest X-ray, the cardiomegaly is significantly contributed by RA rather than RV.
Note the relationship between RA volume with RV dysfunction .
Clinical implication of RA size and function
*As on the left side, we have right atrial functional TR , worsening the RV function in vivious cycle.
*We are in the era of TEER and Percutaneous bio prosthetic tricupid valve. Unlesss we assess the intrisic fcuntional integity of right atrium , we may not be able to get optimal out come .
*More recently consensus conference on pulmonary hypertension, which was adopted by ESC, has included RA area with a cut off 18sq.cm (It should have been volume ideally) as an indicator of PH (Fischer L,. Respir Res. 2018 )
Final message
For cardiologists, LV will remain the emperor of the Heart. RV and also LA are also receiving enormous attention. Sadly RA is hidden in a discriminatory blind spot. It is ironical that we sort of give more respect, even to IVC and SVC flows than RA dynamics. I think we may have to pay a price for it if we continue to neglect it. It may take more time for us to study the right atrial mechanical function and its ability to overcome the dysfunctional right ventricle (RV diastolic dysfunction).
Reference
1.Aune, E.; Baekkevar, M.; Roislien, J.; Rodevand, O.; Otterstad, J. E. . (2009). Normal reference ranges for left and right atrial volume indexes and ejection fractions obtained with real-time three-dimensional echocardiography. European Journal of Echocardiography, 10(6), 738–744.
2.Sallach JA, Tang WH, Borowski AG, Tong W, Porter T, Martin MG, Jasper SE, Shrestha K, Troughton RW, Klein AL. Right atrial volume index in chronic systolic heart failure and prognosis. JACC Cardiovasc Imaging. 2009 May;2(5):527-34. doi: 10.1016/j.jcmg.2009.01.012. PMID: 19442936.
3.Querejeta Roca G, Campbell P, Claggett B, Solomon SD, Shah AM. Right Atrial Function in Pulmonary Arterial Hypertension. Circ Cardiovasc Imaging. 2015 Nov;8(11):e003521; discussion e003521. doi: 10.1161/CIRCIMAGING.115.003521. PMID: 26514759; PMCID: PMC4629509.