Normal P waves
What are the components of Pwave ?
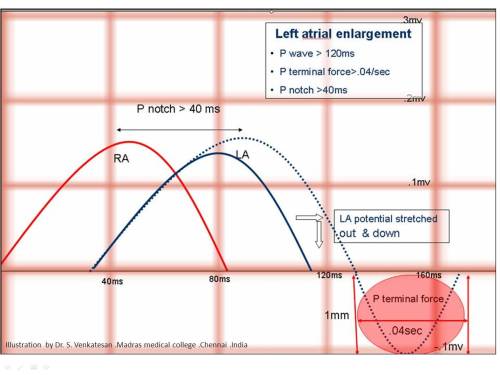
RA component : The SA node depolarises the RA first , so the initial part of P wave represents RA current .After about 40msec the wave front reaches LA and it begins it’s depolarisation .LA component :By the time LA is maximally depolarised the RA already starts its repolarisation.So there is overlap and also a short time lag between these two wave forms . This is very important to recognise as , even if the RA conduction is prolonged in pathology the RA component of P Wave still falls within the LA wave .Hence it is not shown in the ECG and P wave is not widened in RA enlargement. This is in contrast to LA enlargement , when the terminal half of P vector delayed it stretches the P wave wide beyond the normal 110ms .Hence LAE widens the Pwave.
Why P wave becomes taller in RA enlargement ?
In classical P pulmonale , the P waves are tall >2.5mm. It is easy to explain why it not getting wide than why it is getting taller ! The atrial vector has two components .The initial RA vector is directed anteriorly .The main reason for tall p with RAE is due to the anatomical proximity of RA to the chest wall Further ,the Initial atrial electrical dp/dt is steep . Any RA voltage increase is easily picked up by the chest leads and P wave voltage increase and becomes tall. We need to realise LA is not only left of RA its equally posterior of RA. Hence LA enlargement rarely brings (Never ?) it closer to chest wall ,and hence high voltage tall P is almost unheard of with LA . Note , deep negative late P wave activity is typical of LAE , consistent with its posterior location as well its late depolarisation compared to RA)
Why LAE can not produce tall P wave ?
The Left atrial vector which follows RA vector is mainly directed posteriorly and hence inscribe a descending limb of P wave . This causes the P terminal force . So the direction of vector forces and the anatomical locality make a tall & positive P deflection highly improbable in LAE .
*Of course when LAE is huge , where a antero -supero vector from roof of LA may inscribe a positive wave .
What happens in bi atrial enlargement ?
It can have features of both . Tall & wide P waves .
Can RA generate a Q wave ?
Yes . When RA assumes a huge size , especially if the RV is also at high pressure as in severe PHT or valvular PS a q wave is generated in the lead V1 .This q wave is nothing but the intra cavitary potential of the enlarged RA.
What is the difference between atrial enlargement, atrial dilatation, atrial hypertrophy, intra atrial block and inter atrial block ?
The p wave morphology has no specificity to identify the various entities. In any pathology of atrium the first thing that happens is a conduction delay ! It is now realised the bulk of the changes we see in atrial enlargement especially in LAE is due to intra and inter atrial blocks or more subtly conduction delay.
It is obvious , a wide P wave can occur either due to LAE or simple conduction delay .In elderly hypertensive patients atrial fibrosis is more common , one can not confirm LAE without echocardiogram .
A notched P wave can be a very specific sign of inter atrial block .Which is more common in severely diseased left atrium. A notch , slurred p wave is a good marker for impending AF or atrial flutter.
//