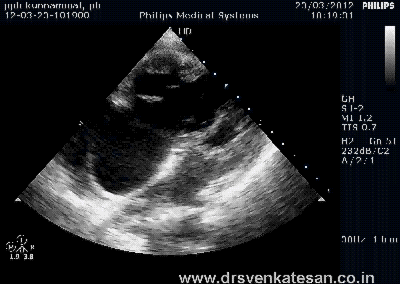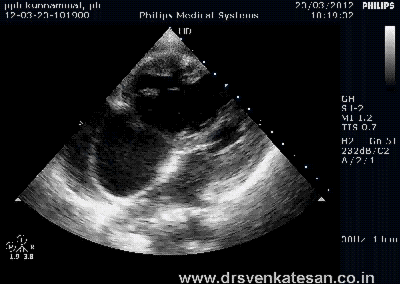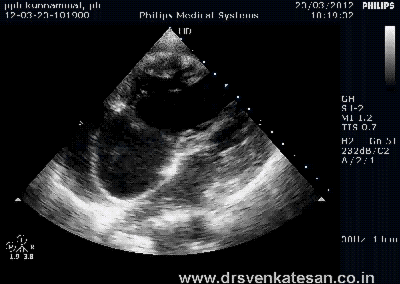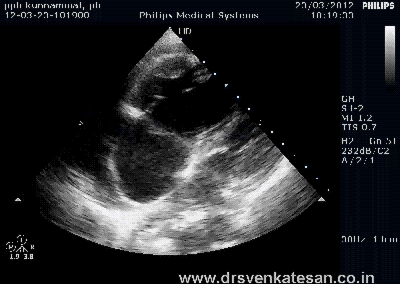
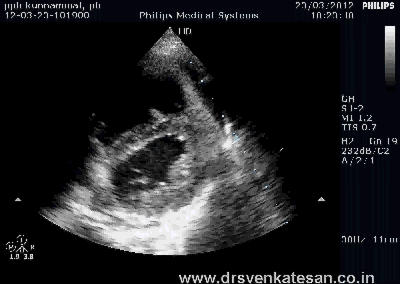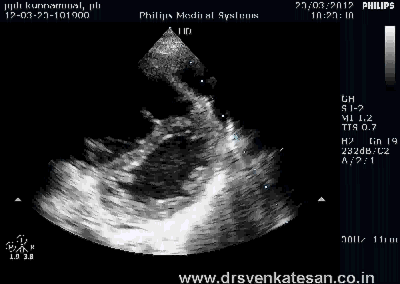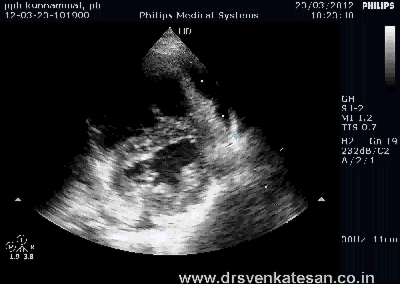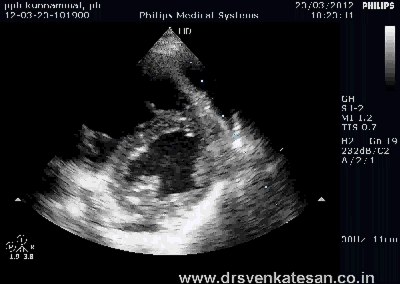
ASD device closure is rapidly gaining popularity . Amplatzer device occluder has become a de-facto standard. Contraindications are declining . More and more young cardiologists want to indulge in this play . Fortunately cost of device is acts as a major deterrent .
The pre procedural evaluation seems to be many fold important than the procedure itself.
- Evaluation of Rims
- Thickness of IAS
- Estimation of size of the defect
- Shape of the defect*
- RA /LA size orifice discrepancy*
- Proximity to Aorta, AV node
- Ruling out fenestrated (daughter defects)
* You ask any cardiac surgeon , How variable the shape and size of ASD can be ? To complicate the issue the LA side may show an entirely different shape and diameter compared to RA aspect. The orifice by itself may travel obliquely.
Currently the thickness of IAS* is not taken into account in device selection . It may be unwise to do so , because the thickness of the rim and its interaction with device determines which direction the device will drag (Homing in ) in the long run .
The potential dead space between the device and the septum can be a late focus for thrombosis. CVAs have been reported following ASD device closure.
Classification with reference to size
ASDs can be small (3 to <6 mm), medium (6 to <12 mm), or large (>12 mm),
What is the shape of Ostium secundum ASD ?
- Round (perfect round very rare)
- Oval
- Irregularity oval
- Irregularly round
- Combinations
How is the orifice orientation with reference to plane of IAS ?
- Horizontal
- Oblique
- Combination of the two
Which is the best method to measure the ASD size ?
- Trans-thoracic Echo
- 2DTrans-Esophageal Echo
- Balloon estimated ASD size in fluoroscopy
- Real Time 3D TEE
- Intra-cardiac Echocardiogram
Currently there is some degree of confusion about utility value of balloon sizing . Opinion differs. A meticulously done TEE may be the winner
How do you tackle an elongated and Oval ASD ?

A large ASD with an adjacent daughter ASD . It is very difficult identify this daughter defect by conventional imaging . Intra cardiac Echo may help . Failure to recognize fenestrated defects especially in the edge can lead to poor device approximation
Con-founders in ASD size measurement.
Stretched ASD diameter. (How much stretch ? )
Systolic vs diastolic ASD size
Practical tips for ASD sizing
Add 2mm to balloon/TEE estimated waist.
TEE may be more accurate than the balloon .
Balloon has a inherent issue of over stretching the ASD and false high diameter.
Waists are often circular in the device We do not have oval Amplatzer device.
Accurate sizing is very difficult to achieve , so which side is better to err ? lesser or over size ?
Dangers of under-sizing
- Mushrooming of the device
- Dislodgment & Embolisation
- Residual shunts
- Thrombosis over metal gutter created by intending device
Dangers of over sizing
- Aortic erosion
- AV blocks
Newer modalities for ASD imaging

Intra cardiac echo and real time 3D TEE will facilitate the ASD device procedures
Image source : Heart 2010;96:1409e1417
Final message
ASD device closure is rapidly gaining popularity . Contraindications are declining . More and more young cardiologists want to indulge in this play . Though more children are getting benefited in this non surgical modality , complications are also increasing .
Small centers should not be allowed to carry out these procedures. Fortunately cost of device acts as a major deterrent . A few centers (one or two per state ) is to be developed for high degree of expertise .
Without mastering the art of TEE never touch the ASD device .
The most critical step in ASD device closure lies before the procedure and . . . it is often outside the cath lab !
Always refer large defects and complex ASDs which are adjacent to Aorta and AV to a good surgeon .Get an operative photograph of the defect and re analyse whether device would have been possible in retrospect .
References
1.An important study about sizing of ASD prior to closure from Sri Chitra Institute . ( This study vouch for TEE for ASD size estimation )
2.Ann Pediatr Cardiol. 2011 Jan-Jun; 4(1): 28–33.
3.Sizing Balloon-Induced Tear of the Atrial Septum
4.http://www.invasivecardiology.com/article/4716?page=2
5.J Teh Univ Heart Ctr 2011;6(2):79-84
6.Echocardiography in cath lab -An Excellent review in Heart
Further reading
Related Post in this site. (ASD closure lagging behind surgery ?)