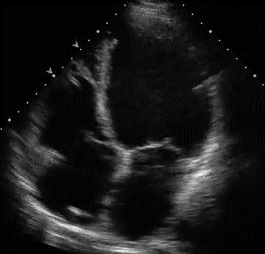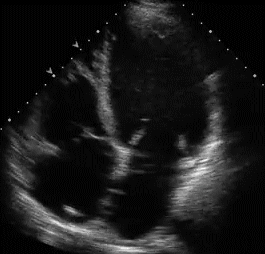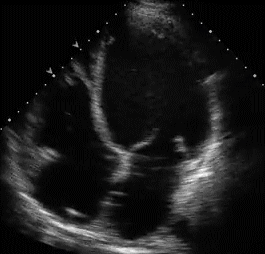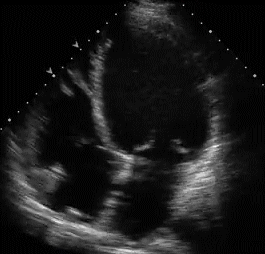
RVH is traditionally categorized into three types . With the advent of echocardiography diagnosing RVH by ECG would appear redundant. Still , it gives vital information about the electro-physiologcal basis of RVH. Knowing different mechanisms of RVH helps us decode regional variations in RVH.
Type A , Type B are easy to diagnose as they fulfill the conventional criteria of tall R in lead V1
Type A RVH occur in severe pulmonary hypertension and critical valvular pulmonary stenosis.
Type B RVH occur in volume overload states like ASD and moderate forms of mitral stenosis.
( Severe MS may cause Type A pattern if RV pressure exceed systemic pressure)
Type C RVH has no classical signs of RVH. Here RVH is diagnosed by proxy . Look for RAE and a vertical QRS axis . ( For all practical purposes RAE will indicate RVH except in isolated tricuspid stenosis.
Type C RVH occurs classically in COPD and in some cases of acute pulmonary embolism .In other- words type C RVH reflects predominantly RV dilatation rather than hypertrophy.
Why Type C RVH is important ?
It is important for two reasons
- It is basically a masked RVH .
- It mimics Anterior MI
Missing the first one and erring in later both can have major implications in clinical cardiology especially during emergencies.
What is the mechanism of poor R wave in precardial leads in Type C RVH of COPD ?
The fact that poor R wave in precardial leads occur in most cases of COPD (whether or not RVH is present or not) convey an important message.
The lack of R wave progression is probably less to do with rotation of RV than the insulation effect lung . Further, the elongated lungs drags the heart down , and make it more vertical and in spite of RVH tall R is not picked up by v1 v2 .
Unlike primary PAH and critical MS where the RVH can dominate the LV , the quantum of RVH is never huge in pure COPD . However , presence of RBBB could alter the R wave amplitude .
ECG in acute pulmonary embolism
This resembles the type C RVH . The R waves in V 1 and V 2 can not gain the voltage acutely.
The S 1 . Q 3 , T 3 pattern if present indicate the acute RV strain and the resultant RV wall motion defect.
.
Clinical scenario : Practical utility of decoding RVH by ECG ?
A middle aged female came to our CCU with acute dyspnea with tachycardia .
Echo revealed a dilated RA and RV . She had mild TR and moderate to severe PAH (The TR jet measured 3.8m/sec)
The MPA showed a hazy shadow suspicious of thrombus . The patient had no evidence for DVT .
The fellows arrived at a conclusion about a severe PAH but , the etiology was debated.
One is chronic thrombo-embolic PAH . Other groups argued for acute massive pulmonary embolism and resultant PAH.
This raised an important therapeutic issue as one of them wanted to lyse the thrombus , the other argued for simple heparin .The argument continued as the first fellow reminded , presence of RA, RV dilatation is a sign of acute RV strain . The other countered the same as it could be a chronic response to pre existing PAH.
How do you know in an emergency , whether the RA, RV dilatation is new onset or a chronic one ?
In spite of good echocardiogram we were confused . Then it struck to us , ECG would solve our problem . It indeed helped us. She had a tall monophasic R in V1 indicating Type A RVH , which suggested chronic PAH and the thrombus in MPA in all likely hood was a sequel to PAH and not vice versa . A type C RVH would have voted in favor of acute pulmonary embolism.
Meanwhile a CT pulmonary angiogram report was available . It showed a small thrombus in MPA and LPA with no clearcut perfusion defects ruling out acute pulmoanry embolism . The thrombus was probably de-nova in- situ thrombus due to PAH.
Final message
It may appear funny for the present day cardiologists to waste so much time to analyze the RVH by surface ECG . But please remember ECG remain the only simple and cheap investigation that transmit live data from the heart instantly .Most importantly unlike other imaging modalities ECG data do not vary with person who records it !
Reference
A very good referen from Basic and Bedside Electrocardiography By Romulo F. Baltazar