There are many organic causes of mitral regurgitation. ( Ischemic , degenerative , valvular , cardiomyopathy etc.) It is not rare for pure electrical events to result in valvular regurgitation. A 70year old man with SHT presented with palpitation and exertional dyspnea .He was later referred for Echocardiography. Echo revealed LVH with intermittent MR and moderate LV dysfunction.
His ECG looked like

Ventricular ectopic recorded in bi-geminal rhythm
His echocardiogram showed

His echo showed randomly timed mitral regurgitation was detected .See the Doppler MR jets below.
We know ventricles are integral part of mitral valve apparatus .Hence it wouldn’t be a surprise to note abnormally timed ventricular contraction can have a major impact on mitral valve function.
When ventricles prematurely begin to contract ( As during VPDs) it interferes with opening of mitral valve. In other words every VPD technically imparts a sort of diastolic dysfunction !
VPDs occur in which part of cardiac cycle ?
VPDs occur either in early or mid diastole . Thank fully VPDs can not occur in systole . (Refractory period )
What would be the status of mitral valve at times of VPDs?
Though it depends upon the timing of VPD , generally it interrupts the rapid inflow period of diastole .
In fact , it converts the cardiac cycle from diastole to a partial systole or a combination( fusion ) of diastole and systole ! *

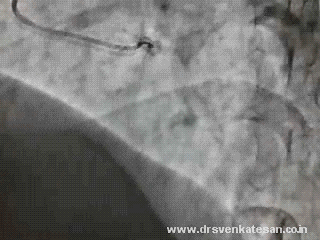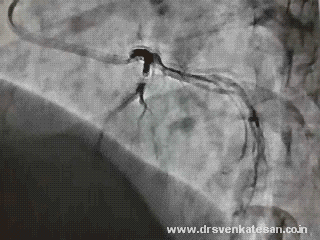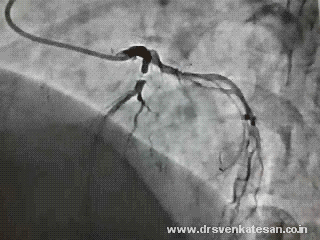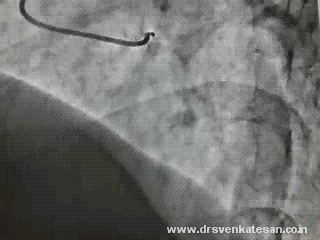
More MR jets are visualised than LV filling waves . Note the some of the E waves are sandwiched between two MR jets. ECG gating should have made this image more interesting .Any way , we have good MR jets to time systole nicely
* Is that a funny imagination ?
During diastole , if LV suddenly begins to contract instead of receiving the blood , what will happen ?
VPDs are such a common arrhythmia , we rarely wondered , it can have a dramatic consequence in a any given cardiac cycle .While the cardiologists think too technically their patients observe with shrewd sense and tell us clearly what they feel is actually a missed beat !
(Yeh . . . how simple they describe the complex hemo-dynamics of missing diastole !) .They also tell us , next systole is felt as big thump as palpitation . (Post VPD potentiation )
Just imagine , if a patient has multiple VPDs with different coupling intervals that fall in different location of diastole also interspersed with sinus beats , how chaotic would be the the mitral filling .
This is what is recorded in the above patient with multiple random MR jets .
Why all VPDs do not cause MR ?
The timing is critical .We know all VPDs do not generate a powerful contraction to cause MR. Atrial fibrillation, Prolonged PR intervals , heart blocks , critically raised LVEDP all can influence the trans mitral gradient . In fact these situation can result in an entity called diastolic MR that would be discussed later.
Can VPD induced MR be referred to as diastolic MR ?
When VPDs occur in diastole , it interrupts the diastole and a new systole begins. In any particular point of time there will be leak into the LA if the mitral valve is open .This is technically a new systole but in true sense it is the diastole of the previous beat . I wonder , whether VPD induced MR may be referred to as one form of diastolic MR. Of course , this MR can spill over to true systole as well .
This also makes sense (Non !) as many of the VPDs do not open the aortic valve , hence technically we can’t call the phase reset by all VPDS as a true systole !
What is the effect of VPDs on pulmonary venous flow ?
Left atrial cannon waves can occur that can elevate PCWP .This is the prime reason for resting or exertional dyspnea in these patients. Some may get a paradoxical relief during exertion as exercise suppress VPDs which are frequent at rest.
If VPDs can seriously interfere with mitral valve function , why they are often considered benign ?
VPDs are well tolerated* as long as the LV function is intact. If VPDs are associated with LV dysfunction it can initiate a vicious cycle of hemodynamic deterioration . Multiple VPDs if left untreated can lead to progressive LV dilatation in a significant population . Hence patients with recurrent VPDS need some sort of follow up. It makes good medical sense to suppress VPDs in the long run. (Of course the available anti VPD drugs are not very safe ! The search for non toxic , ideal drug should go on !)
*”Well tolerated VPDs” in no way means normal physiology. Read a related article in my site. “3 minutes crash course on VPDs”
Final message
VPDs though considered largely benign , can lead to dramatic alterations in the functions of mitral valve , especially in diseased hearts.
We must realise when ventricular ectopic beats occur frequently , it interfere with the both opening and closing of mitral valve.
It is really surprising , the literature is devoid of major studies about the impact of VPDs on mitral valve physiology . . . rather pathology !
Read Full Post »