Posted in Cardiology - Clinical, Cardiology -unresolved questions, Clinical cardiology | Tagged gallop sound, pathological s3, physiological third heart sound, s 3 gallop | 1 Comment »
Third heart sound is a unique heart sound because its perfect physiology to hear it in the young , while the same may denote serious LV dysfunction in patients with myocardial disease.
It is a low pitched early diastolic sound usually correspond to the end of rapid filling phase.The mechanism of genesis of this sound has been debated for many years .(Still I think it is unsettled !)
We know genesis of intracardiac sound is contributed by three factors
- The blood flow
- The valve motion
- The myocardial contractile and relaxyl property
The above three is collectively called cardio-hemic system . When this system vibrates heart sounds are generated .In the genesis of S3 all the three may be important . The only difference is , in physiological S3 the valvular and hemic component play a major role . In pathological S3 the myocardial component has a pivotal role .The distended LV facilitates chest wall impact during the rapid filling phase . It is now accepted LV S3 is generated outside the LV . It was proved elegantly by Shaver et all with sound recording on either side of LV /Chest wall.
It is to be emphasized the mechanism of genesis of S3 is diagonally opposite in physiology vs pathological S3 in some conditions . Rapid AV filling can rarely be responsible for pathological S3 associated with severe LV dysfunction , while chest wall impact can contribute in both physiological as well as pathological S 3 .
One can understand the complexity of genesis of S 3 , as there are too many determinants that contribute in varying degree of acoustics.
Factors determining the intensity of S3 is complex
-
Age,
-
Atrial pressure,
-
Rapidity of flow across the atrio-ventricular valve,
-
Rate of early diastolic relaxation
-
Distensibility of the ventricle,
-
Blood volume,
-
Ventricular cavity size,
-
Diastolic momentum of the heart,
-
Degree of contact (coupling) with the chest wall, thickness
-
Character of the chest wall
-
The position of the patient.
It is ironical, pathological S3 which is a diastolic sound though , still its genesis is largely determined by the systolic function of the heart .This mystery is partially solved as we recognise now , LV S3 is equally common in severe degrees of diastolic dysfunction. In fact , while we were studying the relationship of LVS3 in DCM , it has strongly predicted the presence of severe restrictive pattern in them .
Reference
1.Multimedia of S3
http://www.inovise.com/learn/s3causes.html
2.Importance of S3 in cardiology NEJM 2001 article
http://www.nejm.org/doi/pdf/10.1056/NEJMoa010641
3.Chest wall impact theory of S3 by Shaver
Shaver JA, Salerni R. Auscultation of the heart. In: Hurst, ed. Heart. 8th ed. New York, NY: McGraw-Hill, Inc; 1994:291.
Posted in cardaic physiology, cardiac physiology, Cardiology - Clinical, Clinical cardiology, myocardial disease | Tagged cardio hemic system, chest wall cavity interface and s3, chest wall lv impact and s3, heart sounds, Mechanism of third heart sound, pericardial knock, physioloical verses pathological s3, s 3 and s4 gallop, s 3 gallop, s3, shaver reddy thery of s3, third heart sound, where does lv s3 generated | Leave a Comment »
Mitral regurgitation is one of the most common lesion of rheumatic heart disease .Mechanism of MR in acute rheumatic fever is different from chronic rheumatic heart disease.
Acute Rheumatic fever
The following mechanisms contribute to MR of acute rheumatic fever
- Edema of leaflets (Carey Coombs murmur )
- Valvulitis
- Small verrucous vegetations (See Image )
- Acute LV dilatation in fulminant cardiac failure.
* Note : Acute rheumatic fever in its first episode can never cause stenosis however fulminant the fever may be .There is no acute mitral stenosis .But , during recurrence and reactivation some amount of stenotic process may occur. Still , recurrence and reactivation are more often related to significant MR rather than MS. ( Isolated mitral stenotic lesions rarely give h/o recurrent rheumatic fever )
Chronic rheumatic heart disease
As the mitral valve gets progressively damaged any combination of MS or MR occur .The following mechanism are involved in the genesis of MR. (Pathology of Mitral stenosis is not discussed here)
- Chordal shortening, tethering , pulling , prevent proper co-optation
- Chordal lengthening
- Chordal disruption (Minor > Major )
- Prolapse of either AML or PML (Not both ,unlike myxamatous MVPS)
- Infective endocardits of leaflet
- Perforations of leaflet
- Annular dilatation
- Fibrosis of posteromedial/Antero-lateral pap muscle(Rare )
- Left atrial pathology
* The direction and the width of MR jet is related to the mechanism of MR.
If there is chordal shortening due to fibrosis of mitral valve co -optation plane is altered . The degree of chordal shortening , pap muscle fibrosis (rare) symmetry of chordal involvement determine the MR.
Rheumatic mitral valve prolapse
- This could be more common than we realise.
- It can be true or pseudo.
- True prolapse occur due to chordal weakening or lengthening .
- In chordal disruption the leaflet tips usually become flail
Since rheumatic process fixes the PML first , the AML appear to overshoot the plane of PML and appear as prolapse.(Pseudo )
The sail like AML commonly directs the jet posteriorly and laterally .(Murmur conducted to axilla and back )
It is rare for PML to prolapse in RHD , if it does occur , it directs the jet anteriorly (murmur conducted to aortic area mimic AS !)
It is rare to see a perfect central jet in RHD . presence of Central jet is a good sign to consider mitral valve repair.
Myocardial involvement in RHD.
Even though rheumatic fever is a classical example for pan-carditis , it is surprising to note (Of course fortunately !) how myocardium escapes in the chronic process of RHD.
Is it really true , myocardium do not get involved in chronic RHD ?
Clinical cardiologists rarely discuss this issue. Pathogists indeed have documented significant lesions within myocardium . Involvement of left atrial myocardium and rarely ventricular myocardium in the sub mitral zone can influence the degree of MR
* Even in acute rheumatic fever with fulminant carditis , myocardial involvement is disputed by many ! . My belief is , there will definitely a subset in both acute and chronic forms of RHD , in which myocardium gets involved . In our institute LV dysfunction associated with RHD occur in up to 5 % of RHD population .
Importance of knowing the mechanism of MR
Two aspects appear important
1. Is there a potentially reversible component in pathology so that we can wait before intervention ?
I have seen children referred for mitral valve replacement due to severe MR . In due course MR regress by the time they reach the tertiary center (waiting period included ) At least one child i remember, the MV surgery was canceled due to spontaneous regression MR.
It was later found the MR was more of valve inflammation than degeneration .
* Always think about the possibility of reversible rheumatic MR in every severe isolated MR in children (Do not apply this rule in adults or in combined MS or MR ) Do a ESR, ASO and start an intensive anti inflammatory therapy , aspirin with strict penicillin prophylaxis .With this one can definitely postpone the surgery in few cases and may avoid it altogether !
2. Surgical implication
If we could delineate the exact pathology of MR it will facilitate the repair . Annular reduction and neo chordae etc . Of course ,the surgery could be very difficult in scarred mitral valves , Dr Sampath kumar *of AIIMS New delhi , India would feel other wise !
*A pioneer in mitral valve repair in chronic RHD (See reference 2 )
Questions that need answers
How is balloon/Surgery related injury different from rheumatic process ?
Why is rheumatic mitral vale prone for bacterial infection ?
What is the relationship between extent of aortic valve involvement and degree of mitral valve involvement in RHD ?
Reference
1.http://circ.ahajournals.org/content/94/1/73.full?sid=10599470-3563-4c38-b688-c5fc8c032f96
 2. http://icvts.ctsnetjournals.org/cgi/reprint/5/4/356
2. http://icvts.ctsnetjournals.org/cgi/reprint/5/4/356
Books
There two popular books exclusively for cardiac pathology
Posted in cardiac surgery, Cardiology - Clinical, cardiology -Therapeutics | Tagged acute rheumatic fever, and chronic rheumatic heart disease., mechanism of rheumatic mitral regurgitation, mitral regurgitation, mitral valve pathology, mitral valve prolapse, rheumatic heart disease | 1 Comment »
Sustaining a STEMI may be a pathological end point for coronary artery disease. But , from the management point of view it is actually a starting point for CAD evaluation .Strategies to prevent further cardiac events must be formulated .
How do you manage a asymptomatic un-complicated post STEMI patient* at discharge ?
- Do a sub- maximal symptom limited EST and then discharge.
- Advised to come back after 2 months for a stress test or Perfusion imaging
- Continue with intensive medical management without EST or CAG and monitor only the symptoms
- Advice coronary angiogram in all and decide depending upon the lesions (Pre -discharge CAG )
- I am a modern day cardiologist . This question does not arise . . . as I do only primary angioplasty for all my cases !
( *Please note , this forms the bulk of STEMI population (up to 60 % )
Answer : Your guess is the correct answer!
Why we need to risk stratify STEMI at discharge ?
The morality and outcome in STEMI though appears to be a continuously falling curve , the slope is not linear.
The classical mortality till discharge is about 6-8 %. Between discharge and 3oth day there is 1-2 % additional mortality
At end of first year there is further 2 % mortality. From second year onwards there is an annual attrition rate up to 3 %.
The aim of doing a pre-discharge EST is to do identify ” patient subset ” who are destined to die within 30 days of STEMI. If you schedule the EST after 6-8 weeks one can not prevent these two deaths out of 100 !
( Of course , we assume a prompt revascularisation in those vulnerable would prevent this !). By doing so , we can avoid the bulk of unnecessary PCIs that happen with routine CAG following STEMI.
Pre discharge EST can be done safely within 5-7 days with a symptom limited test (70 % of THR or up to HR of 120 /mt ) This simple test if it is negative can virtually R/O a critical proximal lesion with near 100% sensitivity.
Should we risk stratify patients who have undergone pPCI as well ?
Most of us would love to believe , once pPCI is done to the patient , he reaches a therapeutic end point. But it is not the truth . It is the degree of LV dysfunction , extent of contrary coronary lesion , co existing risk factors and the intensity of medical treatment only would determine the long term outcome.
It is very important to realise the pPCI is aimed at opening the IRA and other lesions are often left alone. So never believe pPCI per se would confer total risk reduction following a STEMI . There is considerable evidence to suggest the opposite may be true at least in high high risk pPCI ,where metals are placed in complex , vulnerable thrombotic milieu. Hence it seems logical to risk stratify all patients after primary PCI (In fact, this population require more vigilance ) .
When will you advice an EST following pPCI ?
It is usually not needed in the immediate discharge phase in single vessel disease which would have been tackled during pPCI.In multi-vessel CAD , where only the IRA was tackled during pPCI ,the same guidelines that of thromolysed STEMI shall apply .Since we know the coronary anatomy already , EST helps us to evaluate the hemodynamic status of non IRA lesions if there are any . While , this is a logical debate , logics has a rare place in medicine . It is ironical , stress test is rarely done even after 6months following pPCI in most centers.
Final message
It is a pity , anatomical risk stratification has squarely beaten the scheme of physiological risk stratification in most cardiology centers . A pre -discharge EST* was a good concept that gave us an idea about the coronary reserve after the ACS. It was a collective wisdom of cardiologists that has hanged this useful concept. It is still more shocking , to note even the scheduled 6 week EST is dropped from the post MI work up in some institutions.
* Many would consider ordering an early EST in STEMI is an act of bravery ! The fear seems to be genuine and most will agree with that. But , please remember a physiological test (Cheapest and simple is EST or a Nuclear perfusion ) should precede CAG in all asymptomatic post STEMI population whenever possible . If EST could not be done prior to CAG for some reason , at least do it following the CAG . It will have an important impact on the downstream decision making which is often an inappropriate PCI !
Posted in Cardiology - Clinical, cardiology -Therapeutics, Cardiology -unresolved questions, cardiology- coronary care, Cardiology-Coronary artery disese, Clinical cardiology, myocardial disease | Tagged heart rate limited stress test, post STEMI est, pre discharge stress test, risk stratification following stemi, sub maximal stress test, symptom limited stress test | Leave a Comment »
Atrial fibrillation is one of the common tachycardias encountered in cardiology practice.In this condition even though atria fibrillates up to 600 times a minute, only a fraction of that reach the ventricles. Thanks to the AV node.It acts like an electrical sink . Hence in most episodes of AF , the ventricular rate will be manageable and hovers between 150-220 .We also know , in the presence of accessory AV nodal pathway there is a risk of 1:1 conduction and result in ventricular fibrillation and risk of sudden death. (Sudden death in WPW syndrome)
A case scenerio
The other day my resident called me to inform about a patient with atrial fibrillation and hypotension .
I told him , to control the heart rate with Amiodarone and I shall come in shortly ,
He replied , the rhythm looked to him like a bradycardia ! I asked him to wait , when I went there , it turned out to be an interesting ECG .
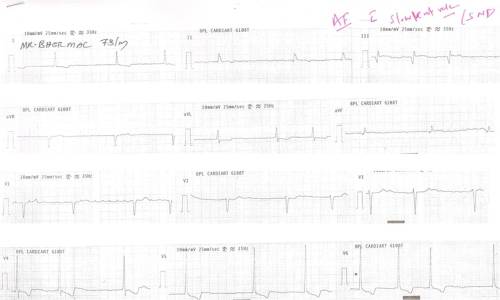
This was a 73 year old man in our ER with a syncope .

Magnified view of lead V 1 and V 2. Note the ventricular rate of 40 /mt even a the coarse f wave are recorded > 300 mt .He had a structurally normal heart .This patient has been adviced a VVI pacemaker .
While atrial fibrillation is primarily a tachycardia , occasionally like the above patient it may present as bradycardia !
How this happens ?
As mentioned before AV node acts like an electrical sink .
How AV node is able to filter out much of the incoming impulses is not clear. This property of AV node is actually the major physiological property of AV node .This is refered to as decremental conduction (The faster it is bombarded with electical stmuli the longer it will take rest !) When this filtering function of AV nodal tissue is too much we call it pathological AV nodal response.Some believe , it is an expression of associated pathological high-grade AV nodal block . Others belive it is simple vagotonia.
Another possibility is it is a sequale to complete AV block and what you witness is nothing but a junctional escape rhythm ( But here RR interval would be fairly regular )
Excessive AV nodal blockers (Digoxin/Verapamil ) can mimic the same picture .
What is the relationship between sinus node dysfunction(SND) and atrial fibrillation ?
AF with slow ventricular response is common in elderly population with sinus node dysfunction.
AF can be associated with SND in two ways
- Atrial disease and sinus node dysfunction is known to occur together . This is not surprising, considering the close proximity they live. SA node is surrounded by atrial tissue in its entire length and breadth .When degenerative and infiltrative disease of atria occur it may trigger a simultaneous SND , as well as atrial fibrillation .
- While another possibility is that AF is a default electrical response to SND . There is some evidence to suggest the atria may release a ectopic escape rhythm which may either degenerate into AF or mainfest a primary AF .
What is controlled ventricular rate and what is slow ventricular rate in AF ?
- This aspect is not well-defined in literature.
- Controlled response generally means HR 70- 90/mt
- Slow ventricular response would be <60 /mt
- Pathological bradycardia is diagnosed with HR < 50 or at any symptomatic slow rhythm .
- Holter or event monitors would help in these situations.
What is the incidence of AV nodal disease in SND ?
AV nodal disease is seen in significant population of SND.(Some series show up to 30 %) .Further , the incidence of new onset AV block increase with every year of follow up ) Reversible forms are commnly due to drugs and electrolyte disorders. The AV nodal disease has another importance as they determine the selction of pacemaker mode .SND with intact AV node function can be managed with atrial based pacemaker ,while ventricle must be paced in patients with AV block or in whom the risk of AV block is high.
Is there a clinical advantage of having some AV nodal disease in AF ?
It may seem so , as long as the AV nodal disease do not lead to severe symptomatic CHB.A slow ventricular rate is a desirable response in patients with angina and cardiomyopathy (especially tachycardic ). While we continue to debate for years about the superiority of rhythm control over rate control , if the AV node chooses to slow down by natural means , ventricles would welcome it with pleasure !
Final message
Atrial fibrillation is primarily a tachyarrhytmia , occasionally it may present as bradyarrhytmia .In this scenario one has to suspect hidden AV nodal as well as sinus nodal dysfunction. ( This entity was also refered to as Tachy brady syndrome ) It is important to recognise this entity because many times dangerous bradycardias have occurred with a single dose of Amiodarone bolus or DC shock . These episodes represent “unmasking effect” of occult AV nodal disease.
Posted in Cardiology - Clinical, Cardiology-Arrhythmias | Tagged AV node in sinus node dysfucntion, complete heart block in af, controled ventricualr response, double nodal disease, sinus node dysfunction, slow venticular response in atrial fibrillation | 5 Comments »
The OAT extension study , a 6 year follow-up study on total occlusion following STEMI has just out in circulation 2011 October , online first . http://circ.ahajournals.org
There were two important conclusions from this study
- Long term follow-up to 6 years confirmed the lack of benefit of routine PCI in post MI total occlusions.
- Inappropriately done PCIs convert stable coronary occlusive disease into potentially dangerous subsets with risk of re-occlusion (Which could very well be an acute coronary syndrome )
The second one is of critical important than the first .In a nut shell , it suggests routine PCI in CTOs could increase the risk of ACS many fold in other wise stable patients.
Final message
This OAT extension study should not experience the same fate of COURAGE and OAT -1 which were successful bitten and buried by most interventional cardiologists.
This time they have to swallow the unpalatable truth ! If they don’t , our patients would be the ultimate losers and
will pay the price dearly !
Personal foot note :
One of my colleague asked me . . . Why am I always after the Interventional cardiology community !
I said , it is not my job to pull down any one group. I am just exposing the irony of “selective usage” or “selective neglect” of scientific data by many of us !
Posted in Cardiology - Clinical, Cardiology -Interventional -PCI, cardiology -Therapeutics, Cardiology -unresolved questions, cardiology- coronary care, Cardiology-Coronary artery disese | Tagged chronic total occlusions following stemi, courage oat 1 oat 2, cto following stemi, inappropriate pci in cto, oat circualtionaha, oat extension trial, oat longterm followmup, oat trial, oat trial new york leon charney | Leave a Comment »
The other day when I was observing my colleague puncturing the inter atrial septum with heavy bore needle during a PTMC procedure the patient was comfortably watching and enjoying the procedure .
Even as multiple wires criss-crossed the left atrium and the balloon hitting the mitral valve repeatedly there was absolutely no pain.
Next day , in an another patient when IAS was punctured it got stained along with pericardium , the patient had severe back pain and procedure was to be stalled temporarily .
What is the lesson learnt ?
The pericardium and the epicardium (same as visceral pericardium ) has rich pain fibers. The above patient who had stain of epicardium had severe pain .
The former patient who had a perfect IAS puncture did not develop pain while the later who had an epicardial track experienced pain.
The same analogy can be seen in patients with myocardial rupture .While sever chest pain is a rule with a free wall tear , it is very rare for patients with ventricular septal rupture to complain sever pain as IVS rupture do not cleave the epicardial layers .It is also uncommon for papillary muscle or chodal rupture to generate significant pain .
What is the difference between epicardium and endocardium in terms of pain fiber innervation ?
Sub- endocardium has less dense nerve supply than sub-epicardium. This is one more reason why isolated sub-endocardial stress less commonly result angina ( Eg Hypertension and sub endocardial strain often silent ) while even minimal irritation or insult of sub-epicardium induce severe pain.
Further , cardiac pain receptors respond differently to type of stimuli .The density of these receptors also vary depending on planes of myocardial tissue .
What are triggers for cardiac pain ?
Any of the following can trigger cardiac pain.The pain receptors in heart are not well developed as that of somatic system.
It is not clear whether the layers of heart has specialized receptors for various sensations.
- Stretch*
- Prick
- Guide wire poke ,
- Needle prick
- Temperature .
- Infection ,
- Inflammation of myocardium , pericardium*
- Pressure injections
- Cardiac ischemia*
* These three factors are responsible for bulk of the cardiac pain . Please note needle prick on the heart is least painful !
How does ischemia generate pain ?
The ischemia of myocytes secrete
Bio chemical
Substance P , prostaglandins, serotonin, adenosine, bradykinin, and other mediators are involved
Neural
Carried by myelinated A-d and unmyelinated C fibers run in the cardiac sympathetic nerves . It is understood ,both the fibers respond to mechanical stretch while Type C fibers also carry chemo signals from bio chemical mediators as well .
Vagus nerve has a major role in carrying afferent signals of pain . It is well known , if pain stimuli is substantial the vaso vagal reflex is activated and bradycardia and hypotension occurs.
How is infarct pain different from Ischemic pain ?
Necrosis of nerve terminal will result in more intense pain and lasts longer .
Clinical examples for stretch induced cardiac pain
- Acute RV/LV dilatation of any cause
- Pulmonary artery/Aortic dilatation
- Pericardial stretch could contribute more in generating this pain
- Mitral valve prolapse (Stretches LV free wall )
Interventional cardiologist should thank god for not innervating the heart extensively . This only allows us to spend hours together inside the patients heart , other wise one would require a general anesthesia for doing a PCI
Does pericardium suffer from ischemia or necrosis ?
Pericardium is not an avascular structure . Pericardium gets its blood supply from twigs of LIMA and phrenic arteries.So there must be some impact of ischemia on pericardium . Since pericardium has rich nerve supply there is every reason to suspect existence of ischemic pericardial pain as well .
But pericardial pain induced by mechanical stretch and inflammation is much more common .While acute pericardial stretch is painful chronic stretch as in slowly accumulating pericardial effusion is a painless event !
Pain relief after CABG
One of the reasons for angina relief post CABG is attributed to the interruption to pericardial nerve supply.
Reference
This 1957 article from circulation still rules cardiac pain literature . http://circ.ahajournals.org/content/16/4/644.full.pdf+html
http://www.annualreviews.org/doi/full/10.1146/annurev.physiol.61.1.143
Posted in cardiac physiology, Cardiology - Clinical, Cardiology -unresolved questions, cardiology-Anatomy | Tagged cardiac pain, pain receptors | 1 Comment »
V wave is one of two positive upstrokes seen in JVP. Physiological “v” wave is due to atrial filling and reaches the peak at late systole , while pathological ” v” waves are often due to tricuspid regurgitation . It is a mid systolic wave .It is a fusion of “c”and “v” waves .
Here is a patient with dilated cardiomyopathy with severe tricuspid regurgitation who presented with prominent neck veins.
there is no difficulty in identifying the v wave . Careful acuity will reveal a sharp “a” wave as well !
How to measure the amplitude of v waves ?
In JVP, there is a baseline oscillating column . Individual wave spikes occur over and above this baseline . Hence technically there should be two measurements , but we take only the top most part of the oscillating column.
What is the indirect evidence for tall v waves ?
Physiologically “y” descend is integral part of v wave (In fact , “y” descend can be referred to as down stroke of “v” wave ) .For every tall “v” wave there must be a prominent “y”descent . (Probably constrictive pericarditis is an important exception ! )
If “y” descend is not rapid but shallow one can suspect two conditions
- Tricuspid stenosis
- Significant RV dysfunction
How to differentiate v waves from a waves ?
“V” wave is a passive filling wave hence it raises slowly , has relatively shallow summit and occurs in mid or late-systole . “A”waves are due to active contraction of atria . It is a sharp pre-systolic wave . One practical way to recognise “a” wave is , it never stays in the eye , it just flickers. If your eye sees a sustained wave for more than a fraction of moment it can not be “a” wave ! Another point that may be useful is “a” is taller than “v” in right atrium .
Reference
Click below to hear the murmur of TR (Courtesy of Texas heart institute )
http://www.texasheart.org/Education/CME/explore/events/upload/HSPS13_TricuspidInsuff.mp3
Posted in cardiac physiology, Cardiology - Clinical, Clinical cardiology | Tagged cv waves, jugular venous waves, jvp, tricuspid regurgitation, v waves in jvp | Leave a Comment »
Proximal LAD lesions require specific and early Intervention.Hence we need to know what exactly we mean by proximal LAD disease.Unfortunately , it means different things to different cardiologists .There is no dispute regarding the origin of LAD since it begins with bifurcation point .The problem comes with this question !
Up to what distance LAD can be termed as proximal ?
- Bifurcation to “First diagonal” of any size
- Bifurcation to “First Major diagonal”
- Bifurcation to “First septal” of any size
- Bifurcation to “First major septal”
- Bifurcation to “Any major first branch ” (Either septal or diagonal )
Answer : I think 4 is the correct answer . But many believe 5 can be correct as well !
Why there is confusion in the definition of proximal LAD ?
This is because the first branch of LAD itself is not a constant one . It can either be a septal or uncommonly a diagonal.
It should be noted , the septal and the diagonal branches neither respect seniority nor follow a hierarchy .The first diagonal may be diminutive while the second or third diagonal may be major one and vice versa .Further , there can be a trade of in length and caliber of septal and diagonal branches .This phenomenon is also common between diagonals and OMs . All these confound the picture .
Cardiologists even though they are primarily physicians they are pro-anatomy like surgeons when it comes to coronary interventions .
In the strict sense , we need to differentiate a lesion from being physiologically proximal or anatomically proximal !
Is there a proximal LAD equivalent ?
There are three situations this can occur .
- Some times a lesion by definition may not fit in as proximal LAD but physiologically few major diagonals will arise after the lesion.
- Other situation is , LAD lesion may be mid or distal but a major first diagonal may be diseased , making it equivalent to proximal LAD in terms of physiology.
- A mid LAD with a large OM lesion which is running in the D1 territory
Final message
It is ironical millions of cardiology interventions happen for proximal LAD lesions every year without even proper understanding of what we mean by it ! Youngsters are argued to ponder over this issue whenever they indulge in such cases for revascularisation!
Reference
Text books differ in their definition about proximal LAD. Currently , the SYNTAX scoring system has defined the coronary segments in a practical way.
http://www.syntaxscore.com/index.php?option=com_content&view=category&layout=blog&id=1&Itemid=32
Definition from SYNTAX
Posted in cardiac surgery, Cardiology - Clinical, Cardiology -Interventional -PCI, Cardiology -unresolved questions, cardiology- coronary care, cardiology-Anatomy | Tagged distal lad, first major diagonal, first major septal artery, first septal vs first diagonal, LAD, major minor diagonal, mid ald, proxiaml lad means what, proximal, proximal lad, s1 d1 om1, second diagonal, second septal, waht is the defintion of proximal lad | 1 Comment »
If we think we have unraveled all the mysteries of human coronary blood flow we are sadly mistaken . Most cardiac physicians spend their prime life in opening the obstructed coronary arteries playing a role of coronary plumber.
Like any plumber , it is not going to be one time job and our patients would have to hire their services periodically . Many times it turns out to be a 108/ 911 call as well !
Unfortunately , hem0dynamics of coronary blood flow do not follow the principle of water flowing across a domestic pipeline.The most dramatic difference between the coronary blood flow and water pipe is , in the later , as the water is being ferried across the house , neither the building nor the pipe contracts (Unfortunately all our understanding , derivations and research were based on simple physical modules of hydrodynamics in a static delivery system )
Pressure flow relations especially in biological system is not simple. Since our foundations on principles of blood flow is based on this simplistic model , every assumption could be proven wrong. This is what is happening now . Nothing seems to work in a learnt manner.
A patient with 100% occlusion walks comfortably without damaging his muscle.While an other patient would develop cardiomyopathy even if the occlusion is gradual and incomplete ! Hemodynamic logic tells us blood flows from high pressure to lower pressure zones like a water fall !
But coronary waterfall is not a simple and smooth affair. It is not a free fall , even as the water falls there are pumpy interruptions .When these pumpy ride occur even in physiology one can imagine the pathological states , when the coronary artery is blocked , the myocardium is scarred and the systemic blood pressure fluctuates .
While every organ welcomes the systole , as they are fed with blood during this time of cardiac cycle . Heart is only organ which sacrifices its own blood flow during this phase as the systolic contraction interrupts the blood flow .
Determinants of coronary blood flow
What we learnt over the years has been too simplistic. It is not the patency of vascular system that matters. The coronary micro vasculature, the metabolic demand, the neuro humoral regulation etc. For most cardiologists the epicardial patency or stenosis remains the only relevant issue
The reality is much complex to comprehend
- The coronary perfusion pressure
- Coronary flow reserve
- Coronary wave forms
- Sub endocardial vs subepicardial flow ratio
- Effect LVH on myocardial flow
- Coronary venous tone and arterial ischemia.
Now, we have an entirely new concept which proposes (Rather proven concept !) the integrity of myocardial contraction and relaxation on the coronary blood flow. This land mark paper in circulation has identified six wave forms of coronary blood flow This include 4 positive waves and two negative waves
Questions need to be answered
During diastole myocardium relaxes . Only if the myocardium relax optimally the compressive effect of systole on coronary coronary micro vasculature is reversed , intra coronary resistance falls so that coronary blood flow can occur smoothly. We do not know whether diastolic dysfunction would affect the diastolic coronary filling waves jeopardizing the coronary flow.
Myocardial viability is important for one more reason , in the distribution of coronary blood flow .A dysfunctional muscle can not receive and inject the blood deep into sub endocardium (Note this becomes important when revascularising severely dysfunctional segment )
Does myocardium has a calf muscle analogy and behave like a powerful intramuscular perfusing pump .
A breakthrough concept from Davies et all in circulation . These are not new ( Buck -Berg ?)thought about this decades ago . The interest is rekindled in recent years , as complex angioplasties following myocardial infarctions failed to improve outcome and relive symptoms in many .
During primary PCI , no- reflow often denotes a meaning of failed PCI .The issue involved is hydrodynamics of intra myocardial blood flow .The following article partly answers the issue underlying no re flow .http://circ.ahajournals.org/content/113/14/1721.full.pdf+html
Young physicians need to spend more time in basic cardiac sciences . Lest, what we do in cath lab blindly will become a laughing stock ! We have to go back to the golden years of research in cardiac physiology (1960 -1970s) . Mastering coronary angioplasty may increase the blood flow up to the myocardium , but pushing the blood beyond the muscle requires more sense and effort .
A simple hemodynamic model based on physical principles alone is a greatest error we make in cardiac science . * Further, human heart muscle is not only influenced by the quantum of blood it receives but to the great extent the content of blood.The blood caries all the ill effects of systemic diseases and damage the vessels and muscle .The interaction between the blood and the muscle is never an issue in the pure physical labs .( Even animals misbehave !)
Posted in cardaic physiology, Cardiology - Clinical, Cardiology -Interventional -PCI, Cardiology -unresolved questions, Clinical cardiology, myocardial disease | Tagged coronary blood flow, epicardial vs endocardial blood flow, sub-endocardial blood flow, subendocardial blood flow, subepicardial blood flow | 1 Comment »