Posted in acute coronary syndrome, atherosclerosis, platelet function | Tagged platelet adhesion and aggrgation and fatty meal briyani, platelet stickyness and dietery fat, platlet anf fat ldl and triglycerides, tgl mediated platlet aggregation | Leave a Comment »
What are the mechanisms of cyanosis in cyanotic heart disease ?
Most of my fellows have difficulty in answering this question. (It is not the lack of knowledge though !) In my view ,cyanosis can occur , by six different modes
- Reduced pulmonary blood flow with some form of anatomical obstruction in RVOT with a communication between ventricles (TOF physiology ) , atria or both
- Reduced pulmonary flow with obstructive pulmonary vasculature (Eisenmenger physiology )
- Wrong way origin ( RV to Aorta/LV to Pulmonary artery ) : Transposition physiology
- Simple mixing of arterial and venous blood channels within the atria ,ventricle or great vessel without RVOT obstruction .This, in fact can causes increased pulmonary blood flow (Technically left to right shunt ) and still there is cyanosis (These are called as Admixture lesions ) It is also to be noted some of the admixture lesions (Truncus, DORV,etc ) the mixing takes place only during systole , while TAPVC,Common atrium, Tricuspid atresia* admixture is more complete as it happens during entire cardiac cycle.
- Isolated Right to left shunt are very rare ( Pulmonary AV fistula , SVC to LA )
- Complex combination of first 4 (Like bi-directional shunting , TGA combines , AV canal defect , with varying degree of pulmonary obstructive disease) Note : TOF and Eisenmenger are physiologically mimic each other , the only difference is site of resistance to pulmonary flow. RVOT vs Lung vasculature )
* Essentially Atrial admixture is more complete than when it happens at ventricular or great vessel level
For advanced readers only
Now, is it possible for “Net” left to right shunt to result in cyanosis ?
Yes*.Very much possible. The bulk of this group is referred to as admixture lesions with certain caveats.There should be an obligatory mixing without contribution from RVOT obstruction or raised PVR( *Please note theoretically admixture can either be right to left or left to right shunt )
All pure admixture lesions are in fact net left to right shunts. (TAPVC, Single ventricle , Common atrium , Common AV canal ,Truncus, ) This is the group we have been traditionally calling cyanosis with increased pulmonary flow.
Its may also to be noted with surprise some admixture lesions often has less intense cyanosis than other forms as long as pulmonary blood flow is normal and the lung does its job perfectly .
*Please note Isolated classical left to right shunts , ASD, VSD, PDA can never cause significant cyanosis unless there is reversal of flow .However ,many Eisenmenger physiology show net Left to right shunting only ( 1.2-1.5 : 1 or so ) but with a definite right to left component .What we call as typically bi-directional shunt .
How can cyanosis be minimal even in some cases of single ventricle ?
- Even though there is single ventricle , there can be preferential (favorable) streaming of right heart blood flow without gross mixing .
- As discussed before good uninterrupted pulmonary blood flow will make the cyanosis less intense .
Is single ventricle with PS admixture lesion or TOF physiology ?
Though single ventricle in isolation is an admixture lesion, when it has associated RVOT obstruction it ceases to be admixture by definition as mixing is augmented by the obstruction rather than by simple mixing.The complexity could be understood in certain situations where admixture lesions like common AV canal go for raised PVR .Here the various quantum of contribution to cyanosis is mind boggling. (Original admixture, augmented by RVOT resistance, differential mixing at atrial and ventricular level , hypoxia at lung level due micro pulmonary AV fistulas in grade 4 heath Edwards etc )
Role of streaming in Admixture lesions
Streaming is selective flow of venous blood into PA and arterial blood into Aorta even in the presence of large septal defects. Favorable streaming implies good systemic saturation. Unfavorable streaming would mean PA saturation more than aorta.(It should be noted streaming and good admixture don’t go together. If good admixture has happened there can’t be any streaming and vice versa)
Streaming is common in which situations?
Inspite of absence of IVS, streaming has been noted in some cases of single ventricle with minimal cyanosis with good saturation in Aorta.
Streaming in TAPVC has some unique features.
Fetal circulation has certain preformed pathways. IVC blood deflects to LA through ASD/PFO .SVC blood preferentially enter RV-PA. In Infradiaphragmatic TAPVC where it drains into IVC highly saturated PV blood may stream into LA thorough ASD and reach LV nd result in higher Aortic saturation.(This is in contrast the classical type of TAPVC draining into RA with little favorable streaming and hence O2 saturation equilibrates between PA/Aorta.)
In Supra cardiac TAPVC that drains into SVC or coronary sinus the streaming is unfavorable as it may preferentially cross tricuspid valve and enter PA making the saturation higher than Aorta.
Streaming is less common in which lesions ?
In common atrium and TAPVC draining into RA streaming is less common.In tricuspid atresia streaming is almost impossible as TV is non existent and this ensures complete mixing in the atria and hence cyanosis is likely to be severe.
Can TOF behave like an admixture lesions ?
Technically yes.If the RVOT obstruction is minimal ,(What was called then as pink Fallot ) We haven’t understood this entity properly for so long. Atleast I was baffled to read when J.K Perloff mentioned in his book during my DM fellowship days, that TOF can manifest with predominant left to right shunt with little or absent cyanosis.
The aortic override in TOF facilitated by large malaligned VSD make it a sort of admixture situation as RVOT resistance is too little to offer any resistance, (rather it welcomes more blood from left side ! ) So , should we call it simple VSD physiology , admixture physiology or just acyanotic forms of TOF ?)
Key points
Though admixure lesions are discussed separately , bulk of them actually represent cyanosis with increased pulmonary blood flow situations.
The net pulmonary blood flow is much more important than the quantum of admixture in determining the degree cyanosis
Finally , one should appreciate there can be combination admixture lesions with obstructive RVOT components . (Tricupid atresia+Pulmonary stenois )
Further reading
An excellent review article on this rare topic of admixture physiology
Posted in admixture lesions in cyanotic heart disease, cardiology -congenital heart disease, Tetrology of Fallot | Tagged admixture lesions in congenital heart disease, can left to right shunt cause cyanosis ?, mechansim of cyanosis, mechansim of cyanotic heart disease, what is admixture lesion | Leave a Comment »
On this special day , wishing all the readers and followers of this blog an energetic, creative , insightful and of-course a happy new year 2016 !
Just wanted to share the 2015 annual report of this site with the readers.
Posted in Uncategorized | Tagged 2015 annual report dr s venkatesan | 1 Comment »
Metformin is one of most commonly used oral hypoglycemic drug listed by WHO as an essential anti diabetic drug. .It is a biguanide which blocks the hepatic gluoneogenesis . Since lactate is the major substrate for the process of gluco-neogenesis , excess of which spills into blood .Lactate is swiftly cleared by the normally functioning kidneys .Metformin is completely excreted by the kidney. Hence in patients with compromised renal function (or when contrast agents compete with Metformin in renal excretion ) high levels would not only cause lactic acidosis (> 5meq), it can also aggravate contrast induced renal injury resulting in a downward hepato -reno-metabolic spiral.
Though the incidence of Metformin induced Lactic acidoss is low , the outcome can be bad , hence the concern. The European society of urogenital radiology has provided clear cut guidelines regarding Metformin usage when contrast agents are being used.
What can be done in emergency situations
- Since the risk of lactic acidosis is very low , in case of emergency situations Metformin need not be discontinued prior to contrast usage.However it need to be stopped for 48 hours from the index procedure. (Fortunately Metformin is a dialysible drug that can be removed in case of toxic accumulation.)
- Consider alternate mode of Imaging if renal function is really concerning
Reference
2. http://ccforum.com/content/pdf/cc12886.pdf
A good article from drug review Contrast induced nephropathy and metformin
Posted in contrast induced nephropathy | Tagged contrast induced nephropathy and metformin usage, eusr guidleines for contrast and metformin, metformin and coronary angiogram, should we stop metformin in all ? | Leave a Comment »
Wall motion defect , in patients after CABG is fairly common.These defects are difficult to interpret as the mechanisms can be multiple.Though the commonest wall motion defect appears to involve the interventricular septum. it can occur anywhere in antero-lateral zone.
The mechanism attributed is the effect of pericardiotomy , which surgeons as we understand leave it open after grafting .This can cause lack of localised ventricular interdependence and results in a a brisk septal movement (bounce )It is an indirect effect .
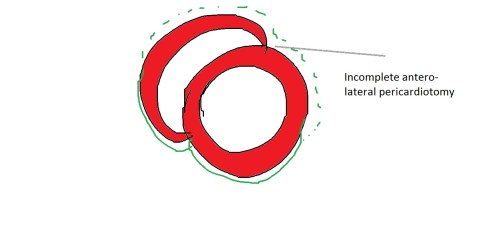
Note the, wall motion defects are confined to the exposed areas of the heart during cardiac surgery .In short axis echocardiography it correlates anywhere between 9 to 3 O clock position. Though interventricular septum is not covered by pericardium in the true sense , there is a indirect bounce effect over IVS due to interference with anterior ventricular interdependence .
More commonly a direct wall motion defect in the 12 to 3 O clock position in short axis is seen .This can closely mimic true wall motion defect as pericardial adhesions can tether these segments. Careful observation is warranted.Myocardial thickening is the key differentiating feature.
What is the physiological impact of these wall motion defects ?
It is generally considered benign (It is !) .Though in echo it looks awkward and suggest desynchrony. The real issue is , it can mislead the echocardiographer to errors in calculation of that universally sacred parameter called EF %
Importance of knowing pre existing wall motion defect.
This has to be reviewed with old reports as it can wrongly create a new wall motion defect de-crediting the surgeons.
New pathological wall motion defect.
Of course it can happen due to peri-operative ischemic insult or infarct . However , It need to emphasised transient wall motion defects are common post CABG due to apparent hypoxia.This seems to be more pronounced with on pump surgeries than off pump .(Expected though) In my opinion, 2-4 weeks cooling off period is required before a meaningful assessment of wall motion post CABG.
Late pericardial reactions and localised constrictive features has been reported.
Disappearance of wall motion defect : How common ?
Any disappearance of WMA is welcome . It happens rarely though . Some of the post ACS population (Both STEMI and UA/NSTEMI) can experience this , as they could harbor zones of myocardial segments afflicted by ischemic stunning rather than true necrosis , that might disappear.
Posted in CABG, Cardiology -unresolved questions, Echocardiography - LV dysfunction | Tagged echocardiography following cabg, paradoxical wall motion defect in ivs septum cabg, post cabg wall motion defect | Leave a Comment »
Inferior STEMI is as common as Anterior STEMI .Unlike the anterior STMI which auto localises to LAD , inferior STEMI has to be fixed either RCA or LCX.
Following ECG features help localize Inferior STEMI .
- ST elevation in lead 3 > lead 2 suggest RCA (Not always true )
- ST depression in lead V1,V2,V3 strongly suggest LCX. (More objectively the sum of ST depression in V1, 2 , 3 divided by sum ST elevation in 2,3, AVF , if less than 1 indicate LCX. Or simply ST depression V3 > Lead 3 indicate LCX.)
- ST depression in lead 1 indicate RCA
- ST elevation in lead V6 strongly suggest LCX
Finally , and most importantly RV infarction as documented by ST elevation in V4R almost always localises the lesion in proximal RCA.
Role of Echo
If ECG features are not clear , a rapid bed side echo has a very good localizing value. To fix RCA look specifically for wall motion defect between “6 to 8” O-clock position .It corresponds to infero basal septum that is invariably supplied by RCA. For LCX involvement concentrate on “3 to 6” o clock position.
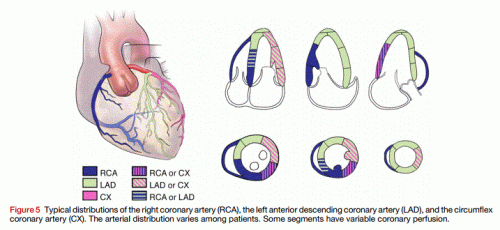
Image source and courtesy http://www.aseuniversity.org
Which has better outcome RCA or LCX STEMI ?
- Though RV infarction does not occur with LCX , incidence of MR is more with LCX and can be truly troublesome. This probably negates the potential advantage of “protected RV” in LCX STEMI.
- Since LV lateral free wall involvement is extremely rare with RCA STEMI , it has a lesser impact on LV function while LCX STEMI can give a double blow to LV (MR and LV dysfunction)
- On the down side ,coronary artery spasm and thrombus load are more with RCA .
Interventions in RCA is fairly straightforward ,while acute LCX PCI has some issues . Apart from technicalities of intubating the posteriorly curving LCX ,realistically it involves fishing in troubled waters , as we need to cross the left main , likely physical contacts with LAD ostium , which is the sole supply chain for the injured and ischemic LV myocardium . Meanwhile , If RCA is the culprit , its a well cordoned crime scene where one can spend time liberally and fix the lesion.
Final message
It is easier to localisethe culprit artery in inferior STEMI ,but its a tricky to predict outcome .Both can be troublesome .It depends on dominance of the RCA/LCX ,proximal nature of lesion, the number and caliber of OMs, and PLVs and RV branch .However, it remains a fact LCX STEMI has a overall turbulent course.
Posted in echocardiography, Infrequently asked questions in cardiology (iFAQs) | Tagged echocardiographic segments in lcx vs rca, localising inferior stemi rca vs lcx | Leave a Comment »
Co-dominant coronary circulation is defined as , when posterior crux of the heart receives twigs from both right and left system making this water shed area with advantage of twin innervation.They essentially supply inferior and posterior aspect of both left and right ventricle including the posterior aspect of interventricular septum.

Image courtesy modified from :http://www.meddean.luc.edu/lumen/meded/mech/cases/case1/image4.JPG
Traditionally inferior and basal aspects of heart are perceived (wrong tough !) as less important than anterior surface of heart.Infero posterior MI can be extensive and cause significant LV dysfunction and poor outcome. Longitudinal function (AV grooval velocity) and Mitral valve function is critically dependent on posterior circulation.
Is there an advantage for co-dominant circulation with reference to ischemic mitral regurgitation ?
Obviously ,one would expect there is some advantage in co-dominant circulation when ACS occurs either LCX or RCA.It could theoretically protect against development of MR as posterior papillary muscles could receive supportive twigs from its companion.
However , there is a caveat .The antero-lateral papillary muscle normally has twin blood supply from LAD(Diagonal ) and LCX (OM) . But in co-dominant circulation this pap muscle is at risk of becoming single blood supply as the dominant RCA has a trade off with OM with its large PLV branch. It is likely in co-dominant circulations if LAD is the culprit outcomes are likely to be worse.
Final message
A rare study involving more than 200,000 patients which specifically addressed this issue of dominance and outcome , threw some surprising findings. In concluded PCI outcomes with left or co-dominance has a worse outcome than Right dominant system.
Reference
2.Papillary Muscle Perfusion Pattern A Hypothesis for Ischemic Papillary Muscle DysfunctioPaolo Voci, Federico Bilotta, Quintilio Caretta,Circulation. 1995; 91: 1714-1718
Posted in Uncategorized | Leave a Comment »
Verdict ?
Only complicated or high risk STEMI, would require immediate anatomy based management. Please note, this population at worst is never beyond 20 % of all STEMI. Hence more than majority of patients can be managed effectively without CAG.
My reasoning tells me,though knowing the coronary anatomy appear vital , it is rather the physiological impact of those anatomical lesions that will determine the outcome. So,post STEMI, if at all , we need to investigate, it should be about the adequacy of the over all blood supply to left ventricle.This is done by a pre or post discharge sub maximal stress /nuclear test .If it’s negative with a good exercise tolerance CAG will never be required as any critical flow limiting lesion ( that would require intervention )is excluded with near 100% surety.
Postamble :Try asking any neurologist , how often they demand to know cerebral arterial anatomy for managing stroke ? You will get a real surprise answer !
Posted in Uncategorized | Tagged fibrinolysis vs priamry pci, indications for coronary angiogram following stemi, priamry pci vs thrombolysis, revascularisation for cad, stemi guidelines, stemi management controversy, timing of coroanry angiogram in stemi, when to do coronary angiogram stemi ? | 2 Comments »
Holter monitoring is the Initial test for all those with documented syncope (or Pre syncope ) with suspected cardiac arrhythmia .It is a 24 hour ambulatory ECG monitoring , expected to pick up any electrical abnormality and its correlation with the resultant symptom if any. Though the test looks attractive , the diagnostic yield is far less. (About 10%) .The reason being the episodes can be rare to be missed by 24hr sample time. We have extended Holter (48hr) , Event monitors , Loop recorders and implantable devices that can record ECG for extended periods.(18 Months ,Reveal Plus Medtronic) that improve the yield up to 45%.
One common issue that often confuse us while reporting Holter is, the pauses that occur during day / night .
What is the significance of these pauses * ? Nocturnal vs Daytime
Pauses are obviously significant when the patient is awake . It is generally accepted pauses more than 3 seconds during day time (ie Heart rate of < 20/mt ) is significant . This is logical , as pauses more than that, is expected to cause syncope ( or atleast pre-syncope ).The problem comes when you document pauses more than 3 seconds without any symptoms . Then this difficult question comes up ,At what degree of pause syncope occurs ? How is that some persons mange even prolonged pauses with just giddiness.(Good overall vascular integrity and tone ! )
We know such pauses are especially common during sleep. How does the brain react when pauses occur during sleep ? as there is no question of fall as such and loss of muscle tone is non existing.
*Please note ,when we say pause we mean only Sinus pause , Pauses due to AV blocks are very significant
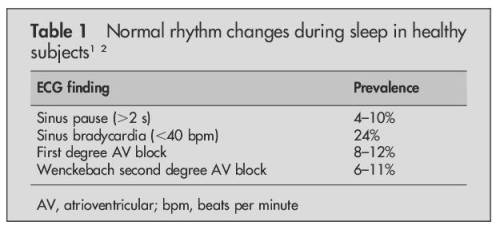
Source : Brodsky M, Wu D, Denes P,et al.Am J Cardiol 1977; .
Dramatic pauses during sleep do occur
There has been prolonged pauses reported during sleep without fatality . A 35 second nocturnal pause resulting in seizures has been documented by implantable recorders.(Mairesse 2003)
Causes for prolonged pauses
- Sinus node dysfunction
- Obstructive sleep apnea
- High dose beta blockers therapy
Final message
Most bradycardic episodes during sleep are benign.This is due to depressed autonomic control during sleep. Holter interpretation is primarily done with awake rhythm data in most individuals .So, empirically shall we fix a 5 second pause as significant during sleep ? We don’t know.While this may seem applicable even with structural heart disease , one may be vigilant while interpreting the nocturnal pauses in this population .
Caution
** Please note, all these rhythm monitoring extravaganza is meant for people with equivocal symptoms .Patients with well documented syncope with ECG features suggestive of cardiac rhythm disorders would never require these tests and go for pacemaker straightaway.
Reference
Posted in Uncategorized | Leave a Comment »
What are the determinants of dissecting path in Aortic dissection ?
Aortic dissection is taught to us as a dramatic cardiac emergency where the blood enters one of the planes of aortic wall and travels in a random way . The wrong way blood instead of flowing within the lumen invades the vessel wall .(Vascular Tsunami ?) It may (or may not) leave the aorta at a distance resulting in various combinations of true and false lumen. Much like a tsumani its also triggered by an energy releasing blood pressure spikes hitting on the weakened aortic wall rupturing the Intima. While acute dissection are often dramatic chronic dissection can be more subtle clinically.
Apart from the site of entry , blood pressure , condition of aortic vessel wall , there seems to be an invisible force that direct the dissecting tract.How it spares or compromises the arch vessels in selected few , as it travels down remain a mystery . If we can predict and track the plane of dissection by any means with computational hemodynamic models , that will help us plan strategies. Beta blockers are used to reduce the shearing pressure , and emergency surgery is required in many type A dissections.
Do we see a “mini” Interventional opportunity here ? To arrest or direct the dissecting tracts into less benign zone. Shall we deploy an emergency metal ring barrier just proximal to aortic arch in Type A or just above renal arteries in type B to prevent vital organ compromise ? This procedure can be done fast , instead planning a elaborate endovascular intervention which is logistically difficult in arch vessel dissection .This could also act as a bridge to definitive surgery. (Can we compare this with bush fire fighting which are tamed by c0ntroled artificial fire lines and thus avoiding spread to residential areas ! )
Posted in Cardiology -unresolved questions | Tagged aortic dissection, plane of aortic dissection, tracking arch dissection | Leave a Comment »








