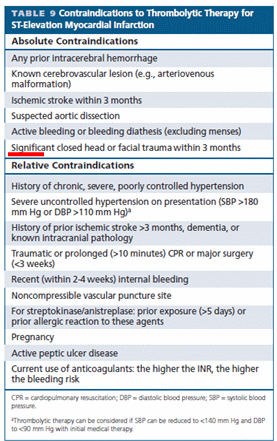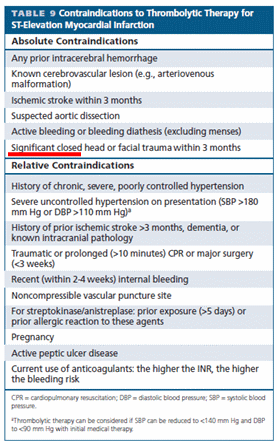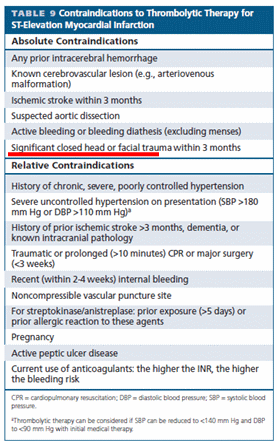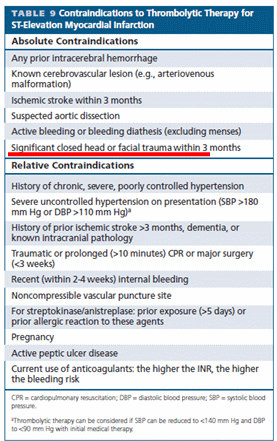
Aortic dissection is a unique cardiac emergency that tests our collective understanding of vascular anatomy and pathology .It poses the ultimate challenge to the expertise and wisdom of both cardiologists, and surgeons.
It’s all about freezing the Time
The philosophy of management swings between near-total Inaction* in some (As in most Type B & few Type A as well ) to “No holds barred” approach in others. (In most Type A and few Type B).
*Read it (also) as medical management that includes powerful Aortic pulse attenuation therapy with beta-blockers ( Unfortunately medical management is considered as Inaction by many current generation cardiologists)
Advanced Aortic Imaging is the key
A rapidly focussed TTE usually confirms the diagnosis.TEE might be used but carries a small risk of directly aggravating dissection when performed in an unstable patient. Conventional CT provides good (but limited) information, spiral MDCT delineates the vascular anatomy in a more clear way. MRI probably scores over and adds flow dynamics.
(4D Phase-contrast MRI showing slow helical flow in the false lumen and high-velocity flow in the true lumen. Computational flow dynamics will help assess entry point, plane of dissection, calculate false lumen Index, pressure and wall stress in true lumen and Aortic branch compromise etc . Image courtesy. The Lancet Volume 385, Issue 9970, 28 February–6 March 2015, Pages 800-811)
What is Non-A Non-B dissection?
This is a newly recognized subgroup. It’s not a surprise as the imaging modality improved we found the existence of this subset. In Non-A -Non-B Dissection initial tear involves the Arch and threaten to go retrograde A or antegrade B. We also realized there could be an apparently illogical transformation of type B becoming Type A, later on, due to late retrograde dissection.
The incidence of Non A , Non B dissection is about 10% (Ref 1). Here the outcome between surgery and medical is confusingly sitting at equipoise.
The traditional Debaky and Stanford classification didn’t address this subset.Though some other classification Like (DISSECT (Ref 2) /PENN (Ref 3) tried to clarify .
A new classification based on Type ,entry and malperfusion appear perfect.
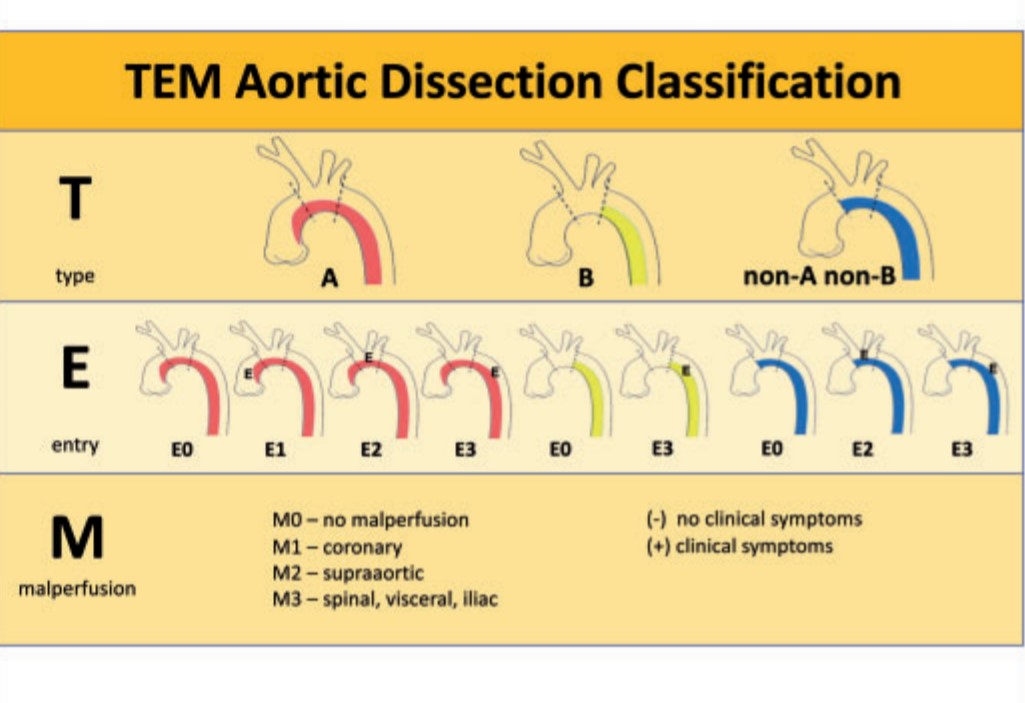
This is a practical classification that uses Stanford as a base model but adds entry point and branch vessel compromise. This is analogous to TNM classification of tumours.(Hans Hinrich Sievers et al)
Where does EVAR stand ?
The management strategy of dissection of Aorta got an important makeover in the last decade (for good mostly ) as interventional cardiologists and radiologists landed in the hitherto to surgical domain with endovascular reconstruction (EVAR) .It is handy mainly in the complicated type B and complicated Non A and Non B . One more viable possibility is the hybrid approach of combining EVAR and surgery in delayed presentation of Type-A.
Single point Principle in Aortic dissection management
Rapid sealing of the entry point and arresting the false lumen progression wherever it is and by whatever means (especially in Type A dissection ) reduces mortality significantly.
Though exit points are important for comprehensive management, one need not waste time to locate and search for exit points. In a specific group of patients, it may not be visible or even absent altogether.
Another critical determinant
Detection and tackling the retrograde dissection and involvement of coronary ostium distortion and damage to Aortic valve , and pericardial invasion is the key to reducing early mortality in Type A dissection
The final outcome in God’s domain
The IRAD and other global registries in Aortic dissection has taught us important lessons. We are continuously learning and the patient outcome is improving.
Still, one unresolved statistical ( metaphysical ) mystery is how to identify? that small subset of patients who are lost because of inappropriately early aggressive Intervention who would have otherwise been pushed into natural survivors of Inaction or less action.
Reference
1.Sievers H-H, Rylski B, Czerny M, Baier ALM, Kreibich M, Siepe M et al. Aortic dissection reconsidered: type, entry site, malperfusion classification adding clarity and enabling outcome prediction. Interact CardioVasc Thorac Surg 2020;30:451–7.
2.Dake MD, Thompson M, van Sambeek M, Vermassen F, Morales JP;DEFINE Investigators. DISSECT: a new mnemonic-based approach to the
categorization of aortic dissection. Eur J Vasc Endovasc Surg 2013;46:175–90
3.Augoustides JG, Szeto WY, Desai ND, Pochettino A, Cheung AT, Savino JS et al. Classification of acute type A dissection: focus on clinical presentation and extent. Eur J Cardiothorac Surg 2011;39:519–22
Metanalysisfor Non A Non B dissection