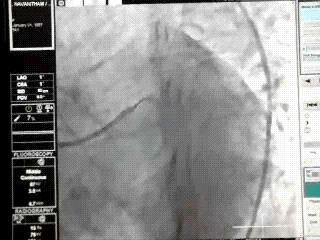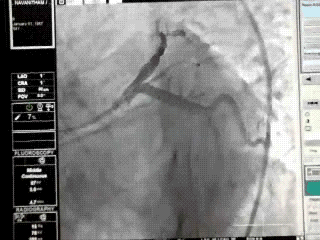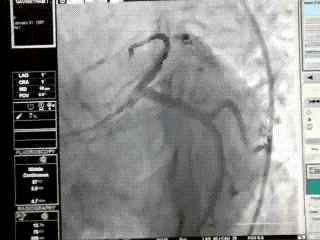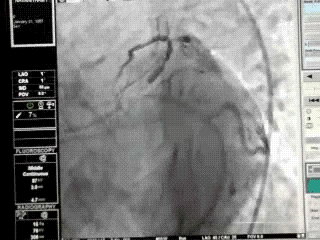
Proximal LAD lesions require specific and early Intervention.Hence we need to know what exactly we mean by proximal LAD disease.Unfortunately , it means different things to different cardiologists .There is no dispute regarding the origin of LAD since it begins with bifurcation point .The problem comes with this question !
Up to what distance LAD can be termed as proximal ?
- Bifurcation to “First diagonal” of any size
- Bifurcation to “First Major diagonal”
- Bifurcation to “First septal” of any size
- Bifurcation to “First major septal”
- Bifurcation to “Any major first branch ” (Either septal or diagonal )
Answer : I think 4 is the correct answer . But many believe 5 can be correct as well !
Why there is confusion in the definition of proximal LAD ?
This is because the first branch of LAD itself is not a constant one . It can either be a septal or uncommonly a diagonal.
It should be noted , the septal and the diagonal branches neither respect seniority nor follow a hierarchy .The first diagonal may be diminutive while the second or third diagonal may be major one and vice versa .Further , there can be a trade of in length and caliber of septal and diagonal branches .This phenomenon is also common between diagonals and OMs . All these confound the picture .
Cardiologists even though they are primarily physicians they are pro-anatomy like surgeons when it comes to coronary interventions .
In the strict sense , we need to differentiate a lesion from being physiologically proximal or anatomically proximal !
Is there a proximal LAD equivalent ?
There are three situations this can occur .
- Some times a lesion by definition may not fit in as proximal LAD but physiologically few major diagonals will arise after the lesion.
- Other situation is , LAD lesion may be mid or distal but a major first diagonal may be diseased , making it equivalent to proximal LAD in terms of physiology.
- A mid LAD with a large OM lesion which is running in the D1 territory
Final message
It is ironical millions of cardiology interventions happen for proximal LAD lesions every year without even proper understanding of what we mean by it ! Youngsters are argued to ponder over this issue whenever they indulge in such cases for revascularisation!
Reference
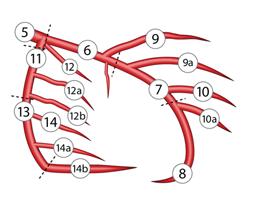
Text books differ in their definition about proximal LAD. Currently , the SYNTAX scoring system has defined the coronary segments in a practical way.
http://www.syntaxscore.com/index.php?option=com_content&view=category&layout=blog&id=1&Itemid=32
Definition from SYNTAX