Radial access for both diagnostic coronary angiogram and PCI has been increasing steadily.Many centres have adopted an exclusive radial approach. Newer , radial specific hard ware is being produced.(Link) .It is surprising , radial approach has gained momentum primarily outside USA (Europe , Japan, India ).The advantages of radial access is primarily , patient comfort, less local site complication.
Prerequisite for radial approach.
The importance of pre procedural Allen test to document dual blood supply is well established but the less appreciated concept is preprocedure radial artery size assessment . It could be as important as Allen test .
The normal diameter of radial artery lumen is 2.4mm (Range 1.8-3.0) .Some population have still smaller radial lumen(India KA.Sambasivam et all mean 1.8mm). Compare femoral artery (8.5mm diameter, 4 times bigger )
Imagine this situation , a 1.65 mm (5 F) diameter catheter trying to enter a 1.7mm radial artery !
Is it not a futile excercise ? Many of the failed radial access is due to radial artery /catheter mismatch .
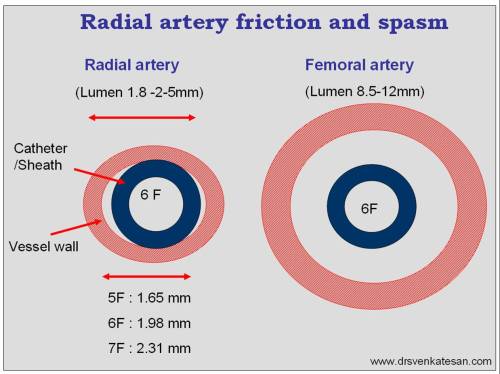
There has been occasions , when the radial sheath is larger than radial artery itself !
So the size of the radial artery becomes vital in planning radial CAG.
What are ways one can estimate the size of the radial artery ?
- Volume of pulse (Still Useful , but can fool us some times!)
- Thick radial walls (Monckeberg,s sclerosis)
- Ultrasound imaging
Radial artery spasm
Radial artery has more medial smooth muscle and further the fibres criss cross the artery. Further , the radial artery is richly innervated by sympathetic nerve terminals.
The major factor that determines likely hood of spasm is
- Pain intensity.
- Amount of free space between sheath and vessel wall
- The frictional force between sheath and artery wall is the powerful trigger for spasm and pain.
What is the biochemical mediators of radial artery spasm ?
It is logical to believe all vascular spasm are due to calcium .But it is not. Calcium blockers have no definite relief for spasm.Nor adrenaline mediated alpha receptor stimulation has a major contribution for RAS. Phentolamine is useful
Is there a objective and quantitative method to assess radial artery spasm ?
Removal of the radial requires some force. Kiemeneij ( Measurement of radial artery spasm using an automatic pullback device. Catheter Cardiovasc Interv 2001;54:437–441.) demonstrated if one require > 1kg force to remove a sheath it correlates with clinically significant spasm.
What are the serious sequel of radial artery spasm ?
Radial artery rupture and radial artery avulsion has been reported when attempting to remove the sheath from spastic arteries
Management of radial spasm
There are two aspects to this problem
- Prevention of spasm
- Treatment of established spasm
How to prevent or reduce radial artery spasm ?
Radial artery is a very sensitive artery .The incidence of spasm can be up to 20% The spasm can be due to
Hardware, technical ,anatomical factors.
Apart from above three factors, the most important is anxiety related .The key principle is , sedating the radial artery is as important as sedating the patient .
Sedating the patient
Explaining to the patient about the procedure can allay the anxiety. It is a fact , the tactile perception of catheter movement in radial route is more than the femoral .In very anxious patients (Some centres use it routinely ) IV sedation (Midazolam)
Sedating the radial artery.
Local anesthesia : Subcutaneous lignociane , though widely used has a drawback.It can aggravate pain, induce spasm , accidental entry into lumen may cause a hematoma .All can potentially make the pulse feeble. So care should be taken in giving minimal lignocaine ( At a specific point needle entry) with a short needle . One should watch , the grace with which experienced radial interventionist give the local anesthetics !
Arterial cocktail
The arterial cocktail consists of combination of Nitroglyceine (200 mcg) , Xylocaine (50 mg) Verapamil (5 mg) an. Heparin(5000 IU). Sodium bicarbonate (4%) is optional to neutralise the acidity of the solution
How to administer ?
Ideally spasmolytic cocktail should be given before the sheath is introduced immediately after puncture . As the drugs has to get in contact with the arterial wall .if cocktail is given after introduction of sheath one of the following may be done.
- Give the drug as it enters the artery.
- Pull back the sheath when injecting the drug.
- Use a side holed sheath.
Technical issues to prevent radial artery spasm
Try to puncture in single prick . If the first puncture is not successful , don’t attempt to cross a spastic radial artery Remember (Unlike femoral ) successful puncturing of a spastic radial artery may be the beginning of a vexing and tiring procedure.So if we have lot of difficulty in getting in , please avoid the procedure and switch to femoral . (Spastic signals may spill over to left hand also !)
Remember unlike femoral cathetrisation , in radial access, getting out the catheter could be more tricky than getting in !
Avoid procedures that would require multiple catheter , guide wire exchange .Complex lesions and in emergencies.( Some experts do Primary PCI through radial !) .Now dedicated radial hardware are available.
Long sheaths have been used in the past. It makes the spastic segment lengthier.Now short sheaths are increasingly used. Long sheaths (20-25cm ) give better suppport during catheter manipulation. Side holed sheaths is a newer innovation. This maintains the blood flow on the sides of sheath and reduces friction with vessel wall .Further, it can deliver the arterial cocktail to vessel wall and effectively prevent or reduce spasm.
Catheters : Use 5 F/6F. Rarely 7f are used.
Guide wires :Hydrophilic guidewire are radial friendly.
How to manage established severe spasm ?
For severe spasm with sticky sheath / catheter
- Increase the analgesia with morphine
- Repeat NTG and Verapamil
- Warm compresses over the forearm
- Never pull with force
- Wait for an hour and try pulling again (Often successful )
Last resorts
An axillary block
Vascular surgery
Reference and further reading
For excellent collection of radial access resources please visit www.radialforce.org
(Much of this blog’s content is based on this article )
http://www.invasivecardiology.com/article/5446
http://meeting.chestjournal.org/cgi/content/abstract/130/4/201S-a