Intra coronary thrombosis is the sine qua non of acute coronary syndrome ( Both STEMI and NSTEMI.) But thrombolysis is the specific therapy in STEMI and is contraindicated in NSTEMI/UA.

Why is this apparent paradox ? What is basic differnce between UA and AMI ?
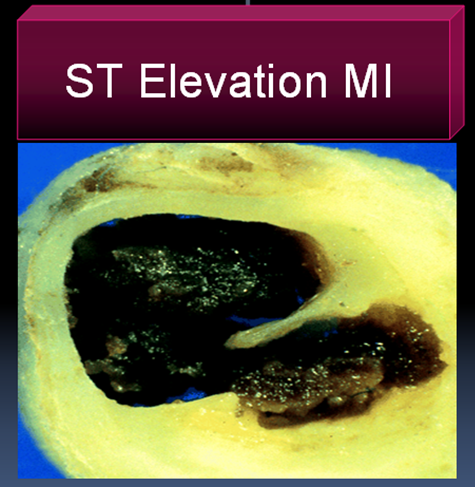 In STEMI there is a sudden & total occlusion of a coronary artery usually by a thrombus with or without a plaque .The immediate aim is to open up the blood vessel . Every minute is important as myocardium undergoes a continuous process ischemic necrosis. So thrombolysis (or more specifically fibrinolysis should be attempted immediately) .The other option is primary angioplasty, which will not be discussed here.
In STEMI there is a sudden & total occlusion of a coronary artery usually by a thrombus with or without a plaque .The immediate aim is to open up the blood vessel . Every minute is important as myocardium undergoes a continuous process ischemic necrosis. So thrombolysis (or more specifically fibrinolysis should be attempted immediately) .The other option is primary angioplasty, which will not be discussed here.
The thrombus in STEMI is RBC & fibrin rich and often called a red clot. Number of fibrinolytic agents like streptokinase, Tissue palsminogen activator,(TPA) Reteplace, Tenekteplace etc have been tested and form the cornerstone of STEMI management.The untoward effect of stroke during thrombolysis is well recognised , but usully the risk benefit ratio favors thrombolyis in most situations except in very elderly and previous history of stroke or bleeding disorder.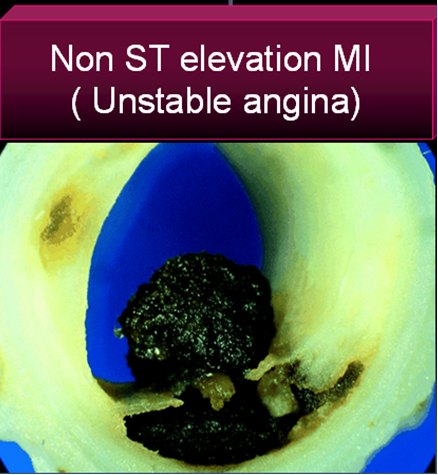
Unstable angina is a close companion of STEMI . Many times it precedes STEMI often called preinfarction angina. During this phase blood flow in the coronary artery becomes sluggish gradually,and patients develop angina at rest .But unlike STEMI there is never a total occlusion and myocardium is viable but ischemic, and emergency salvaging of myocardium is not a therapeutic aim but prevention of MI becomes an aim. It is a paradox of sorts , even though thrombus is present in UA , It has been learnt by experience thrombolytic agents are not useful in preventing an MI .
Why thrombolysis is not useful in UA ?
1.In unstable angina mechanical obstruction in the form of plaque fissure/rupture is more common than completely occluding thrombus. So lysis becomes less important.
2. Even if the thrombus is present , it is often intra plaque or intra lesional and the luminal projection of thrombus is reduced and hence thromolytic agents have limited area to act.
3.Further in UA/NSTEMI since it is a slow and gradual occlusion (Unlike sudden & total occlusion in STEMI) the platelets get marginalised and trapped within the plaque .Hence in UA thrombus is predominantly white . Often, a central platelet core is seen over which fibrin clot may also be formed.
4.All available thrombolytic agents act basically as a fibrinolytic agents, and so it finds difficult to lyse the platelet rich clot.There is also a small risk of these agents lysing the fibrin cap and exposing underlying platelet core and trigger a fresh thrombus.This has been documented in many trials( TIMI 3b to be specific) So if we thrombolyse in UA , there could be a risk of recurrent ACS episodes in the post thrombolytic phase.
5. UA is a semi emergency where there is no race against time to salvage myocardium .Administering a stroke prone thrombolytic agent tilts the risk benefit ratio against it.
6. Among UA, there is a significant group of secondary /perioperative UA due to increased demand situations. Here there is absolutely no role for any thromolytic agents, the simple reason is , there is no thrombus to get lysed.
7.Many of the UA patient have multivessel CAD and might require surgical revascualarisation directly .
So fibrinolytic agents are contraindicated in UA so what is the next step ?
The emergence of intensive and aggressive platelet-lytic agents.
A combination of aspirin, clopidogrel, heparin, glycoprotien 2b 3a antagonist formed the major therapeutic protocol in these patients.Even though these are called antiplalet agents some of them like 2b/3a antagonist eptifibatide, tirofiban, and many times even heparin has a potential to dissolve a thrombus. So technically one can call these agents as thrombolytic agents.
What are the unresolved issues
Even though clinical trials have convincingly shown thrombolytic agents have no use in UA .There is a nagging belief THAT there could be group of patients with UA , still might benefit from thrombolysis as total occlusions have been documented in some cases with UA.This is especially true in peri-infarction unstable angina (Pre & post) as there is a fluctuation between total and subtotal occlusions ) .But bed side recognition of this population is very difficult.
Many would consider this issue as redundant now, since most of these patients are taken up for emergency revascularisations
Read Full Post »